This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Scientists achieve real-time capture of ionization process and subsequent structural changes
Ions are everywhere, from our daily surroundings to the cosmic expanse. As common table salt (NaCl) dissolves into sodium (Na+) and chloride (Cl-) ions in water, it imparts a salty taste. Once absorbed by the body, these ions regulate nerve impulses and muscle movements.
In the sun, plasma—a gathering of ions in the gaseous state—undergoes nuclear fusion reactions, transmitting light and energy to Earth. One of the most noteworthy uses for ions in everyday life is found in lithium-ion batteries, which power devices such as smartphones, laptops, and electric cars.
Consequently, ions play pivotal roles in various facets of our lives, and comprehending the intricate processes, structural attributes, and dynamics of ions remains crucial for advancements in science and technology. However, capturing the ephemeral moments of ion formation and their molecular structural transitions, especially in the gas phase, has proven to be challenging due to experimental complexities.
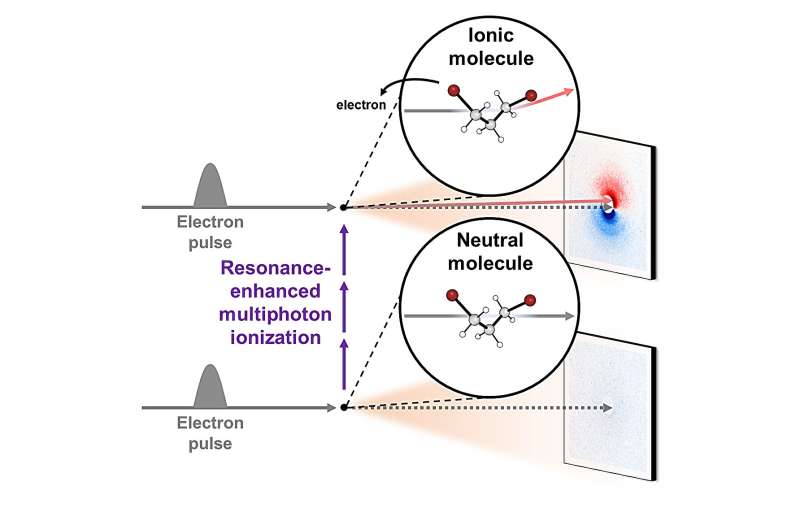
Led by Director Ihee Hyotcherl, researchers at the Center for Advanced Reaction Dynamics (CARD) in the Institute for Basic Science (IBS) have achieved real-time capture of the ionization process and subsequent structural changes in gas-phase molecules through an enhanced mega-electron-volt ultrafast electron diffraction (MeV-UED) technique, enabling observation of faster and finer movements of ions.
Director Ihee's team had a long history of achieving groundbreaking milestones in molecular dynamics, such as the rupture of molecular bonds, the initiation of molecular birth through chemical bonding, and the in-depth exploration of molecular structures at the atomic level across the entirety of a chemical reaction. Now for the first time, they have successfully conducted real-time observations of the formation and structural evolution of gas-phase ions.
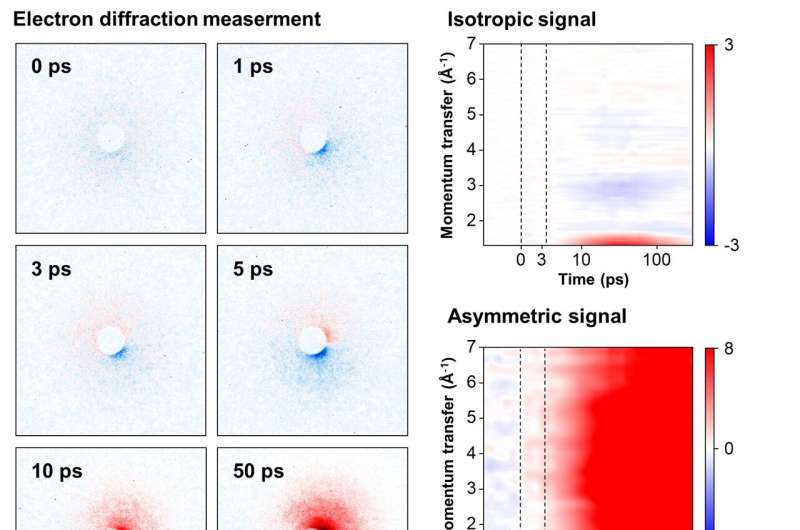
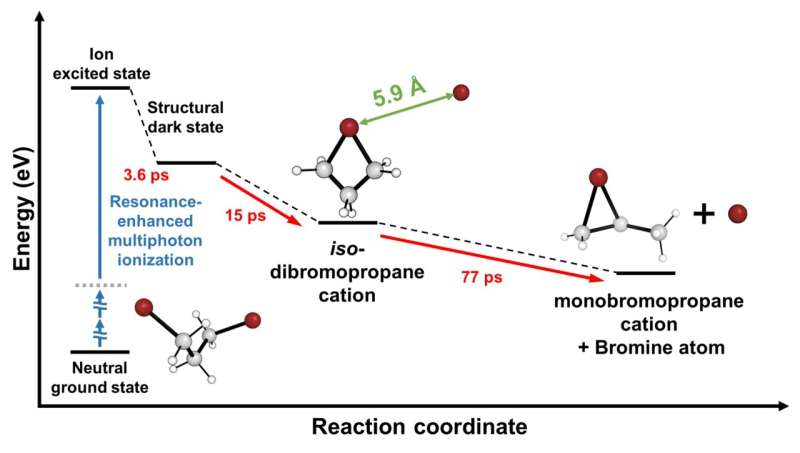
To achieve this objective, the team focused on cations of 1,3-dibromopropane (DBP). Experimental data unveiled a fascinating phenomenon—the cation persisted in a structurally stable state termed the "dark state" for approximately 3.6 picoseconds (1 picosecond equals one trillionth of a second) following its formation.
This new research is published in the journal Nature.
Subsequently, the cation underwent a transformation into an unusual intermediate with a ring structure encompassing four atoms, including a loosely bound bromine atom. Eventually, the loosely attached bromine atom disengaged, giving rise to a bromonium ion characterized by a ring structure comprising three atoms.
Given the high reactivity of ions, observing their existence has posed a longstanding significant challenge. The success of this research hinged on the incorporation of a newly devised signal processing technology and a modeling analysis technique for structural changes. Another important element was the application of the resonance-enhanced multiphoton ionization (REMPI) technique, which facilitated the mass production of specific ions while preventing random dissociation of compounds.
The experimental findings indicated that the generated gas ions maintained a specific form before undergoing sudden transformations, which allowed the IBS team to ultimately elucidate the formation of chemically stable, ring-shaped molecules.
Then, by leveraging the innovative mega-electron-volt ultrafast electron diffraction (MeV-UED) technique, the research team achieved a precise capture of subtle structural changes in ions within the gas phase. This cutting-edge technology offered high-resolution spatial and temporal resolution required for the needs of this research, and it enabled the meticulous tracking of the entire process from the moment of ion generation to subsequent structural transformations.
Being the first to achieve real-time observation of structural changes in selectively generated ions, this study is hailed as a substantial breakthrough in ion chemistry research. This research represents a groundbreaking achievement in the scientific community, marking the inaugural instance of real-time observation of the structural dynamics of molecular ions.
By advancing our understanding of ions in the gas phase, this research yields fresh perspectives across diverse fields, including the mechanisms of chemical reactions, alterations in material properties, and the realm of astrochemistry. The anticipated impact extends well beyond ion chemistry, influencing various scientific and technological disciplines.
Dr. Heo Jun, the primary author, said, "This discovery represents a pivotal advancement in our fundamental comprehension of ion chemistry, poised to profoundly influence the design of diverse chemical reactions and future exploration in astrochemistry."
Kim Doyeong, the first author and a student, shared his aspirations, stated, "Contributing to a study with the potential to lay the groundwork for advancements in basic science is truly gratifying. I am committed to persistent research efforts to evolve into a proficient scientist."
Professor Hyotcherl said, "Despite the remarkable strides in science and technology, numerous captivating mysteries remain in the material world. This research, though unveiling just one more enigma of ions previously undiscovered, underscores the profound secrets awaiting our exploration."
More information: Hyotcherl Ihee, Capturing the generation and structural transformations of molecular ions, Nature (2024). DOI: 10.1038/s41586-023-06909-5. www.nature.com/articles/s41586-023-06909-5
Provided by Institute for Basic Science