This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Novel stem cell culture system could lead to future therapies for a variety of disorders
A new study led by UT Southwestern Medical Center molecular biologists presents a novel culture system to grow both embryonic and extraembryonic stem cells, potentially providing important insights into the genesis of congenital malformations and early developmental disorders.
Published in Cell, the report by an international team of researchers from the U.S., China and Japan outlines procedures for establishing a culture system that, for the first time, permits the simultaneous derivation and co-culture of stem cells from both embryonic and extraembryonic tissues.
"These findings represent significant progress in stem cell and developmental biology through the development of a unified stem cell culture system. The use of this novel system not only deepens our understanding of the interplay between embryonic and extraembryonic cells during embryogenesis but also sets the stage for the creation of more precise models of embryonic development and improved stem cell differentiation protocols," said lead author Jun Wu, Ph.D., Associate Professor of Molecular Biology and a Virginia Murchison Linthicum Scholar in Medical Research at UT Southwestern.
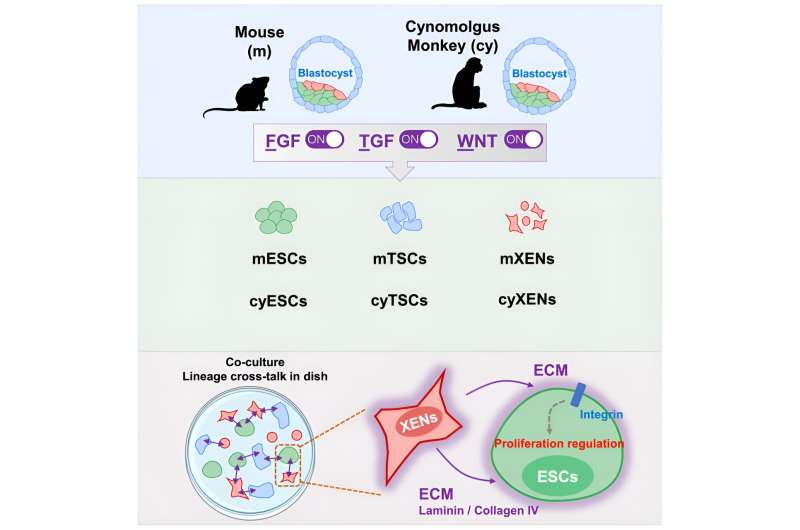
In addition to outlining the new unified growth conditions for embryonic and extraembryonic stem cells, the study reveals critical interactions between different embryonic and extraembryonic cell types, such as the growth inhibition of pluripotent cells by extraembryonic endoderm cells, and emphasizes the importance of cell-to-cell communication during embryogenesis. The study also identifies both common and unique factors involved in regulating extraembryonic endoderm stem cells across different species.
"This fundamental research in stem cell biology holds the potential to provide crucial insights into the genesis of congenital malformations and early developmental disorders. The insights derived from this study lay the groundwork for potentially preventive and therapeutic strategies for such conditions," noted Dr. Wu, a member of the Hamon Center for Regenerative Science and Medicine and the Cecil H. and Ida Green Center for Reproductive Biology Sciences at UT Southwestern.
"Overall, the ability to mimic natural embryonic environments in vitro can significantly advance the study of early human development and diseases, potentially leading to novel treatments and prevention strategies," said first author Yulei Wei, Professor in the College of Biological Sciences at the China Agricultural University in Beijing, who completed her postdoctoral training in 2022 in the Wu Lab at UT Southwestern.
The new findings build upon the Wu Laboratory's earlier work, which has contributed to the generation of novel stem cells for basic and translational studies. Researchers have identified several new types of pluripotent stem cells (PSCs) with distinct molecular and phenotypic features from different species and generated PSC-derived interspecies chimeras and a blastocyst complementation system for the generation of organs and tissues. Most recently, Dr. Wu's group has developed strategies to generate stem cell embryo models for studying peri-implantation development in vitro.
In a 2021 study published in Cell Stem Cell, the Wu Lab devised a culture method that could generate an intermediate, also referred to as formative, state of pluripotent stem cells from the blastocysts of mice and horses. The current investigation discovered that the same culture condition can be used to derive both embryonic stem cells and extraembryonic stem cells, such as trophoblast stem cells and extraembryonic endoderm stem cells, from the blastocysts of mice and primates.
The new study furthers the Wu Lab's research into interactions between embryonic and extraembryonic tissues in early mammalian development utilizing stem cell-based embryo models. Previous work has successfully developed stem cell models of early mammalian embryos, including mouse, bovine, and human blastocyst-like structures (termed blastoids), as well as structures resembling the human peri-gastrulation stage (referred to as human peri-gastruloids).
The findings have the potential to impact research and therapeutic approaches for a variety of diseases and conditions, particularly those related to:
- Developmental disorders: Understanding the coordination between embryonic and extraembryonic tissues can shed light on congenital malformations and developmental disorders that occur during early embryogenesis.
- Placental abnormalities: Insights into trophoblast stem cells that contribute to placenta formation may improve the understanding of placental dysfunctions, such as preeclampsia or placenta accreta.
- Regenerative medicine: The ability to co-culture embryonic and extraembryonic stem cells may enhance tissue engineering and the development of organoids. This could benefit conditions requiring tissue regeneration.
- Cancer research: Since some cancers involve signaling pathways common to stem cell regulation, such as FGF, TGF-β, and WNT, this research may contribute to understanding tumor growth and metastasis.
- Reproductive health: The study may lead to advancements in fertility treatments by improving the understanding of early embryonic development and the implantation processes.
More information: Yulei Wei et al, Dissecting embryonic and extraembryonic lineage crosstalk with stem cell co-culture, Cell (2023). DOI: 10.1016/j.cell.2023.11.008
Journal information: Cell Stem Cell , Cell
Provided by UT Southwestern Medical Center