This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
From waste to wealth: Transforming biomass into humic acid with two-step hydrothermal process via hydrochar
Humic acid (HA) is a macromolecular organic substance that is widely used in agriculture, forestry, animal husbandry, chemical industry, building materials, medicine, environmental protection, and a number of other fields. However, the production of chemical HA in the market relies on non-renewable resources, such as peat, lignite, and coal, which require long periods to form. And the extracted HA yield depends on the type and quality of fossils.
Low-grade lignite and coal usually lead to low HA extraction efficiency. Although traditional composting methods can convert fresh biowaste into fertilizers with high HA content, they are inefficient and take a long time.
Hydrothermal technology can create high-temperature (>160°C) and high-pressure (5–250 bar) conditions in an airtight environment, therefore the hydrothermal treatment of biomass can be considered as a chemical process simulating natural coalification, which provides a renewable and environmentally friendly route to HA production.
Under hydrothermal conditions, hydrochar and hydrothermal solutions derived from biowaste can undergo humification, and alkaline hydrothermal treatment can further improve hydrothermal humic acid (HHA) yield. However, the yield of HA using the alkaline hydrothermal treatment of biomass waste is usually low. The limitation of the low HHA yield is owing to the formation of a large amount of hydrochar with low humification capacity.
So to overcome this problem, in a previous study, a facile two-step hydrothermal humification method was developed to maximize the HA yield by further converting hydrochar to HA using a two-step hydrothermal treatment.
Studies have shown that both the acidic hydrothermal humification of biowaste and the alkaline hydrothermal treatment of the hydrochar contributed to the high yield of HHA. However, there is a lack of research on optimizing acidic hydrothermal humification conditions to further improve HHA yield. In addition, the mechanism of action affecting the humification ability of hydrothermal charcoal in alkaline hydrothermal treatment is not explored.
Researchers from Tsinghua University and Hong Kong Baptist University optimized a facile two-step hydrothermal humification method developed in a previous study. The effect of hydrothermal temperatures and pH on the hydrochar properties and the further influence of the hydrochar-derived HA formation were investigated.
The study, entitled "Two-step hydrothermal conversion of biomass waste to humic acid using hydrochar as intermediate," is published online in Frontiers of Environmental Science & Engineering.
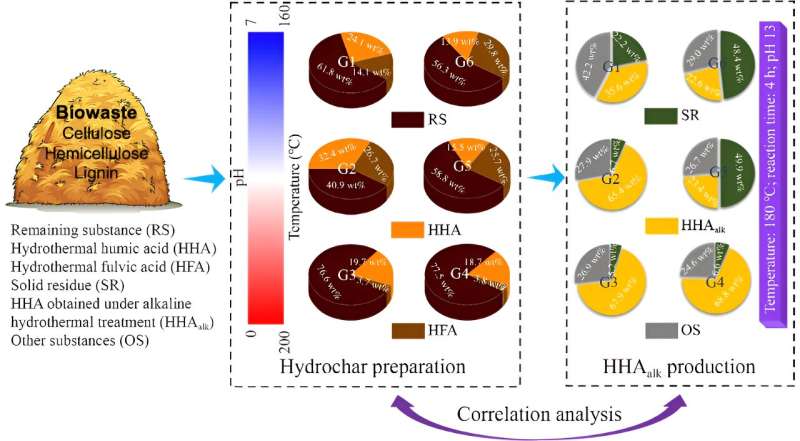
In the study, several biomass-derived hydrochar were prepared and obtained by the research team at different hydrothermal pH values and temperatures. Changes in the chemical characteristics of the hydrochar under hydrothermal conditions were analyzed. In addition, the degree of humification of the hydrochar under alkaline hydrothermal conditions was investigated.
Correlations between the degree of hydrochar unsaturation and their degree of humification were determined.
Their results showed that the hydrochars prepared in the hydrothermal conditions with high temperature and low pH showed high humification capacity, and finally achieved high HHAalk yields. It also revealed that the sources of hydrochar-derived HHAalk were revealed: 1) production in stage of hydrochar preparation; 2) increment under alkaline hydrothermal treatment of hydrochar. The RS in hydrochar played a key role in HHAalk formation.
These results provide a scientific basis for the sustainable treatment of biomass waste and reveal the relationship between biomass-derived hydrochar and its humification potential. This innovative approach is expected to facilitate the production of renewable HA, reducing the dependence on non-renewable resources and providing more sustainable solutions in multiple fields.
More information: Yuchao Shao et al, Two-step hydrothermal conversion of biomass waste to humic acid using hydrochar as intermediate, Frontiers of Environmental Science & Engineering (2023). DOI: 10.1007/s11783-023-1719-8
Provided by Frontiers Journals