This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Q&A: Scientific collaboration paves the way to cleaner technologies for industry

During the nearly five decades of its operation, the European Molecular Biology Laboratory (EMBL) in Hamburg has developed many fruitful collaborations with other scientific institutions located in the Hamburg metropolitan area. One example is the long-lasting collaboration between researchers at EMBL Hamburg and the Center for Biobased Solutions (CBBS) at the Hamburg University of Technology (TUHH), which has recently yielded new insights into the structure and function of a lipid-degrading enzyme found in a microbe adapted to living in extreme conditions. The findings could help improve chemical processes in various branches of industry.
The study is published in the journal Nature Communications.
In this interview, Matthias Wilmanns and Garo Antranikian, who lead respective research groups at EMBL Hamburg and TUHH, discuss how their collaboration developed over almost two decades.
What motivated you to study enzymes from organisms living in extreme conditions?
Garo Antranikian: Throughout my career, I was consistently motivated by the wonders of nature, which never failed to inspire my research work. Collaboration with industry further amplified this creative energy leading to the realization of innovative technologies. We are experiencing a sort of a revolution nowadays—a transition to a circular economy and sustainable technologies that rely on biomass like starch, fats, and cellulose. Biology will play a dramatic role in it by enabling ways to use these resources more efficiently.
Biomass can be degraded to compounds useful for various industries, such as the food and the pharmaceutical industries. It's possible to do this via chemical reactions, but they are not very exact and you get side reactions and contamination. However, in nature, enzymes can carry out reactions very precisely and without producing compounds that are harmful for our environment.
In many cases, enzymes are very specific but complex, so we need to study and understand them in more detail to develop efficient and environmentally friendly enzymatic processes useful for the industry.
For scientists like Matthias and [me], it's extremely interesting to learn from nature. With this knowledge and with artificial intelligence, I think it will be possible to develop completely new nature-inspired enzymes in the future, and make totally new products, such as new bioplastics.
That's why we started working together, and we focused in particular on a lipid-degrading enzyme found in extremophiles—microbes adapted to extreme conditions. Enzymes from extremophiles exhibit remarkable resilience in extreme conditions. Particularly, at elevated temperatures, reactions become both faster and more efficient. This characteristic also allows the development of more sustainable and effective industrial processes.
Matthias Wilmanns: This was actually one of the reasons why I found this project motivating. The structures of enzymes from organisms living in extremely high temperatures are more stable and thus more likely to be solved using structural biology methods.

I had also other reasons, of a more private nature. The organisms adapted to extreme environments come from the most exotic places in the world, and I find it fascinating. For example, the enzyme that we worked on with Garo comes from a microbe isolated from the Solar Lake in the Middle East.
Finally, I have very much enjoyed working with Garo over extended periods of time, especially in recognition of the fact that he has pioneered the field of research with extremophilic organisms.
How did you start working together?
Wilmanns: We first met 23 years ago—in 2000. At that time, I was particularly interested in enzymes from extremophilic conditions and wanted to explore their properties at the molecular level that let them adapt to those conditions.
Antranikian: I'm a microbiologist, so I was interested in microbial physiology—how microbes live, how they make products, how they degrade complex chemical compounds, etc. And Matthias knows the technology to analyze enzyme structure, the interaction of these enzymes with other molecules, and so on.
With interdisciplinary research, you can get much better results, so, we need to work together. And especially in Hamburg, where we're lucky to have so many tools and infrastructures available, Matthias and I had good opportunities to do that.
Wilmanns: That's why I'm very positive about the Science City Hamburg Bahrenfeld, and its potential to stimulate scientific exchange in the metropolitan area of Hamburg. The TUHH in Hamburg- Harburg is another leading hub. This concept has the potential to evolve the city even more as a leading hub for research worldwide.
The research that Garo and I have been pursuing for the last two decades is now also highly relevant for the current EMBL program "Molecules to Ecosystems."

What challenges did you overcome to complete this work?
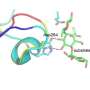
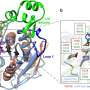
Wilmanns: Our ambition was not just to solve the structure of the enzyme, but to make it useful in terms of possible applications and learn about its mechanisms of function. That's why we aimed to not only solve the structure, but also see how it interacts with lipid molecules as it degrades them.
The ability of lipids to dissolve in water is close to zero, while the enzyme is water soluble. So, there's absolutely no way to get both into a crystal (needed for X-ray crystallography experiments) using conventional methods. We had to find a way around it. It was a lucky break that the enzyme is heat-resistant, because at high temperatures, the lipids mix with the water and the enzyme a little bit—just enough for us to capture them together in an experiment.
What did you learn from this collaboration?
Antranikian: Of course, scientifically, we've learned a lot from each other. But I've also learned to be patient and not to lose hope. And I've learned that from Matthias, because he's more patient than I am.
Wilmanns: For me, Garo opened my mind to research areas I wasn't familiar with before. The collaboration enabled me to learn about the unique research profile of the TUHH and different ways of approaching research, with potential applications very much in the mindset. Through the collaboration, I got to know many researchers with whom I later collaborated.
What advice would you give to other scientists who want to start interdisciplinary projects?
Antranikian: From my experience, I got my best papers when the projects were not well planned [laughs]. This is because when you plan everything and you expect certain results, the level of innovation is very low. You should be very enthusiastic, motivated, and then try to take risks.
Also, you have to try to collaborate with people that you don't naturally expect to collaborate with, who are outside your discipline, and then try to create new ideas. Communication is extremely important, such as interaction with other researchers, institutions, industry etc. Without it, you can't really get innovation.
More information: Nikos Pinotsis et al, Discovery of a non-canonical prototype long-chain monoacylglycerol lipase through a structure-based endogenous reaction intermediate complex, Nature Communications (2023). DOI: 10.1038/s41467-023-43354-4
Journal information: Nature Communications
Provided by European Molecular Biology Laboratory