This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers enhance the function of natural proteins using 'protein Legos'
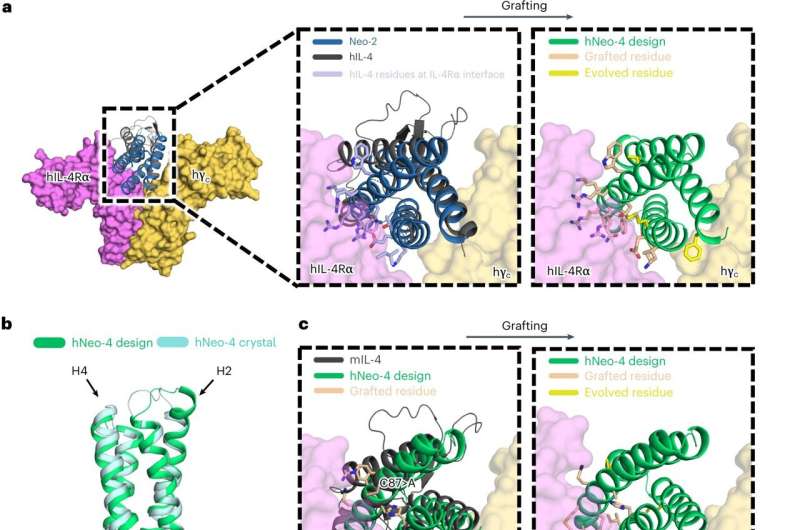
Johns Hopkins engineers have helped develop and characterize an artificial protein that triggers the same response in the human body as its natural counterpart—a breakthrough that not only has the potential to facilitate the design of drugs to accelerate healing but also sheds light on the mechanisms behind various diseases.
The team's research was published in Nature Chemical Biology.
"It's protein Legos, essentially," said team leader Jamie Spangler, an assistant professor of chemical and biomolecular engineering and biomedical engineering. "We know what the different pieces look like, and we put them together in an arrangement that is predicted to look like the protein we're trying to mimic. As far as the body is concerned, this newly created protein is as genuine as the one that occurs in nature."
The synthetic protein, called Neo-4, mimics the function of the natural protein interleukin-4 (IL-4), a crucial player in immune system regulation. White blood cells release IL-4 in response to a range of immune triggers, from allergic inflammation to muscle injuries. IL-4 can then attach to various receptors on cells throughout the body. However, when IL-4 is directly injected as a drug, it can bind to unintended cells, causing unwanted side effects.
"If you give someone IL-4 it just acts on everything," said Zachary Bernstein, team member and Ph.D. candidate in biomedical engineering. "But that makes it difficult for therapeutic use. Neo-4 is more specific and only activates immunologically relevant cells."
Neo-4 attaches to a narrower range of cells than IL-4, a characteristic that the researchers say could make it a promising candidate for future drug development. For instance, a torn anterior cruciate ligament (ACL) is a common season-ending sports injury. Cytokines like Neo-4 have the potential to speed up the healing of torn ACLs and other damaged ligaments and muscles.
"These are computationally designed proteins that behave like proteins in nature but have better properties," Spangler said. "That means we can build these robust, hyper-stable proteins to do whatever we want. The hope is that we can use this mimetic to deliver IL-4 in a way that is safer and more robust than the natural cytokine, which could help with its therapeutic advancement."
Huilin Yang, graduate of the doctoral program in chemical and biomolecular engineering and current postdoctoral fellow at ETH Zurich, contributed to this research.
More information: Huilin Yang et al, Design of cell-type-specific hyperstable IL-4 mimetics via modular de novo scaffolds, Nature Chemical Biology (2023). DOI: 10.1038/s41589-023-01313-6
Journal information: Nature Chemical Biology
Provided by Johns Hopkins University