This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Successful polycarbonate synthesis using the photo-on-demand interfacial polymerization method
As an engineering plastic having high transparency and impact resistance, polycarbonate (PC) is employed in eyeglass lenses, camera lenses, DVDs, automobile headlights and bulletproof glass.
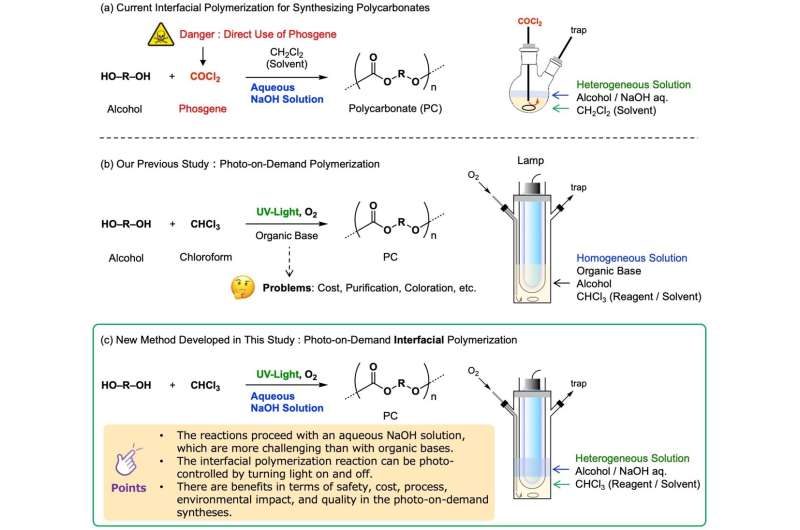
The industrial production thereof is mainly performed by means of interfacial polymerization reacting highly reactive phosgene with alcohol at the interface of water and an organic solvent. However, because phosgene is extremely toxic, a synthesis method not employing it has been actively studied from the perspective of safety.
In recent years, phosgene substitutes (diphenyl carbonate, etc.) has been employed in the synthesis of relatively low molecular weight PC, but for the manufacture of high-grade PC with a large molecular weight, there was no alternative to using the conventionally very-more-reactive phosgene.
With the objective of developing a new chemical reaction enabling the synthesis of safe, inexpensive, simple and low environmental load carbonates, the research group of Associate Professor Akihiko Tsuda of Kobe University Graduate School of Science has worked on the development of an interfacial polymerization reaction by means of the photo-on-demand organic synthesis method developed by that same group.
They discovered that when ultraviolet light was shone on a mixed solution of aqueous sodium hydroxide, chloroform and alcohol (in a three-phase separated state of the gaseous phase, aqueous phase and the organic phase), while oxygen gas is being bubbled through it, a reaction is generated at the interfaces and the target polycarbonate could be obtained at a high-yield. This new method is suitable to the small-scale synthesis of a wide range of carbonate products.
The research team has obtained patents on the photo-on-demand interfacial synthesis method of carbonates in U.S., Singapore, Japan, China, Germany and eight other countries. A paper relating to this study was published in ACS Omega.
The research team succeeded in synthesizing carbonates safely, inexpensively, simply and with a low environmental load by merely shining ultraviolet light on a mixed solution of commercially available chloroform, alcohol and an aqueous solution of sodium hydroxide. Generally, because an aqueous solution of sodium hydroxide breaks down both chloroform and phosgene, and while it would be expected to prevent reactions, surprisingly contrary to that expectation, the reaction was promoted.
Currently, most polycarbonates are manufactured by reacting phosgene gas with alcohol. In the synthesis method described here there is no need to directly handle phosgene, because the required amount of phosgene is generated by irradiating the light to the chloroform solvent.
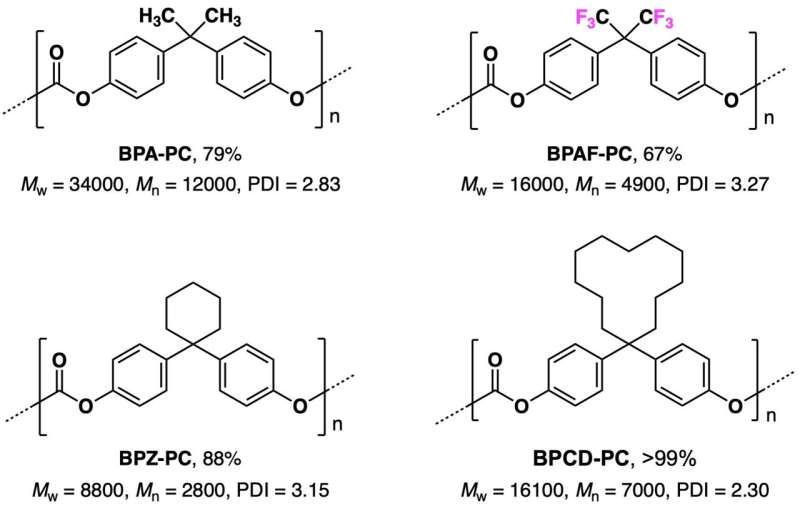
The research team succeeded in synthesizing four types of commercially available carbonate, three types of fluorinated carbonate (used as a phosgene substitute), three types of general use polycarbonate, one type of a specific fluorinated polycarbonate and six types of urea derivatives.
Whereas the conventional method employing phosgene directly is preferred to large-scale production of a small variety of carbonates, this synthesis method is preferred to the production of a wide variety of polycarbonates on a small scale. This could contribute to the implementation of a carbon-neutral and sustainable society with low CO2 emissions, energy consumption and wastes. This study was supported by Adaptable and Seamless Technology Transfer Program Through Target Driven R&D (JST A-STEP SEED Oriented Results).
Phosgene (COCl2) is used as the raw material for pharmaceutical intermediates and polymers. The worldwide phosgene market continues to increase at a scale of several percentage points per year at present, with 8–9 million tons produced annually. However, because it is highly toxic, there are active studies on the development of compounds and chemical reactions which can replace it for safety reasons.
The research group of Associate Professor Akihiko Tsuda discovered for the first time that when ultraviolet light is shone on the chloroform solvent, the photochemical oxidation occured to produce phosgene with high efficiency (Patent No. 5900920).
Furthermore, the research team discovered a synthetic method in which the reaction substrates and catalysts for reacting with phosgene are dissolved in chloroform in advance, and when phosgene is generated by irradiating with light, they react immediately to give the product. (Patent No. 6057449). This method enables the implementation of organic synthesis using phosgene, while apparently not adding phosgene. This research team named that effect "the photo-on-demand organic synthesis method," and succeeded in synthesizing a great number of useful organic chemicals and polymers.
When compared with the conventional direct handling of phosgene, the photo-on-demand organic synthesis method features safety and economy, as well as a low environmental load, and is receiving a lot of attention currently from industry and academia as a next-generation novel chemical synthesis method.
A number of chemical reactions using phosgene are known and used in industrial production. The research team has currently discovered that the photo-on-demand organic synthesis method can be used in most of these chemical reactions, and continues that research, while actively announcing that activity in journal articles, conferences and to mass media.
This study employs a heterogenous solution mixing alcohol, chloroform (CHCl3) and an aqueous sodium hydroxide (NaOH) solution, traversing the three phases of a gaseous phase, an aqueous phase and an organic phase, and succeeded in developing a new photo-on-demand interfacial reaction.
Using this photochemical reaction, the research team succeeded to synthesize carbonates from aromatic alcohols, and polycarbonates (PC) from aromatic diols, enabling a safe and practical scale, high-yield, simple, inexpensive and low environmental load synthesis (Fig. 1, reaction c; Fig. 2). Compared with some of the reactions in a homogenous solution using organic bases developed previously by this research team (Fig. 1, reaction b), there are the benefits that synthesis costs can be reduced, savings can be made in the purification, as well as suppressing the discoloration of the products.
The photochemical oxidation of chloroform to phosgene could be triggered by vigorously stirring the sample solution under oxygen bubbling and irradiating both the gas and liquid phases with UV light from a low-pressure mercury lamp. Because an aqueous solution of sodium hydroxide (NaOH) generally decomposes chloroform and phosgene, it could be expected to prevent the reaction, but surprisingly, it promotes the reaction, contrary to that expectation. It is thought that because the solution is separated into the two phases of the aqueous phase on the organic phase, the decomposition is suppressed, and the reaction is promoted.
It is expected that the alcohol and water competitively react with the generated phosgene in situ, at the interface of the organic phase/aqueous phase and the aqueous phase/gaseous phase, generating carbonate. The highly volatile alkyl alcohols diffused in the gaseous phase did not cause the reactions, but carbonates were obtained at a high yield from commercially available aromatic alcohols and diols having electron-donating and electron-withdrawing substituents.
Most of the industrial production of polycarbonate is currently performed by the interfacial polymerization method using phosgene directly (Fig. 1, reaction a). The method described here avoids the direct handling of that dangerous phosgene, and is an innovative and practical chemical reaction enabling the synthesis of polycarbonate by a photo-on-demand interfacial synthesis reaction of chloroform and alcohol.
More information: Akihiko Tsuda et al, Photo-on-Demand In Situ Phosgenation Reactions That Cross Three Phases of a Heterogeneous Solution of Chloroform and Aqueous NaOH, ACS Omega (2023). DOI: 10.1021/acsomega.3c04290
Journal information: ACS Omega
Provided by Kobe University