This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Rise of the RNA machines: Self-amplification in mRNA vaccine design
The next generation of self-replicating mRNA vaccines could have fewer side effects such as fever, headache and fatigue. This would partly result from the application of artificial intelligence in vaccine development. This is the prediction of Gorben Pijlman, virologist at Wageningen University & Research (WUR), and colleagues, published in Trends in Biotechnology.
mRNA vaccines won the race in the early approval of COVID-19 vaccines, but they also cause side effects such as fever, headache and fatigue. Improvements are needed to maintain their leading role in infectious disease control. According to Pijlman, the possible solution lies in a next generation of self-amplifying mRNA vaccines and the use of artificial intelligence.
An mRNA vaccine contains a piece of genetic information (RNA). That information causes the body to produce a characteristic protein of the virus: the spike protein. Parts of this protein are recognized by immune cells in the body. This causes the immune cells to spring into action when an actual infection occurs.
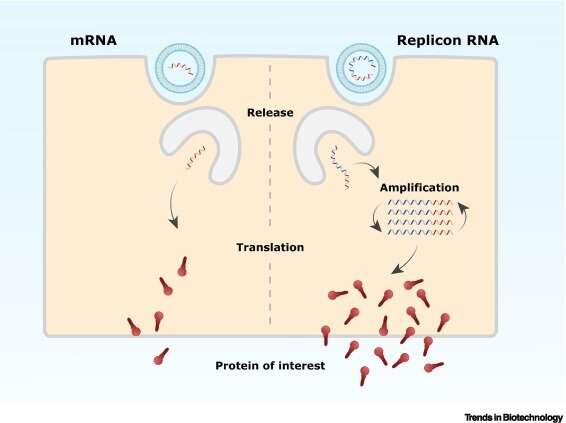
"We think that a building block needs to be added to these mRNA vaccines," says the WUR researcher. "A copy machine that causes the delivered RNA to replicate in the body; these so-called replicons, or copied RNA, ensure powerful responses with few side effects after immunization with a single, low dose. Replicon vaccines have already been used for animals for some time, with great success. In India, vaccines that work this way for humans are now also permitted."
Copy machine
The copy machine, or polymerase (protein), is currently derived from a virus. Pijlman expects scientists to be able to make it themselves in the future, for example with the help of artificial intelligence. "With the implementation of artificial intelligence in protein design, we expect it will be possible to design small, efficient polymerases based on the structure and shape of all viral polymerases in public databases," he explains.
But these future AI replicons may not be covered by current legislation on genetically modified organisms (GMOs). They are not from or associated with any particular virus family. In other words, these advanced "RNA machines" will not be subject to GMO legislation and/or registration procedures that could restrict or at least delay the market authorization of replicons.
Pijlman states, "Nevertheless, it remains important to mitigate any risks of replicons to humans, animals and the environment. Clear legislation is needed to do this properly; in any case, technology is moving faster than regulations can keep up."
Pijlman points out that replicons are not only deployable against infectious diseases: "Replicons could also play a role in inducing an immune response against cancer. In addition, they could protect against multiple diseases or different variants of a disease. A lot is possible in the future."
More information: Jerome D.G. Comes et al, Rise of the RNA machines—self-amplification in mRNA vaccine design, Trends in Biotechnology (2023). DOI: 10.1016/j.tibtech.2023.05.007
Journal information: Trends in Biotechnology
Provided by Wageningen University