This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Model microchip that uses human iPS cells could reduce need for animal experimentation
Animal experimentation may not be a thing of the past just yet, but work on human iPS cell technology may someday grant emancipation for lab mice and other species.
Renal proximal tubules are an essential part of our kidneys' ability to reabsorb vital substances into the bloodstream, such as albumin, before the conversion to urine.
However, in order to pursue accurate testing and other applications, researchers have needed a quantitative evaluation system that simulates the function of these tubules; existing methods could only evaluate epithelial cells using animal experiments and cultures.
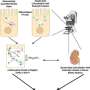
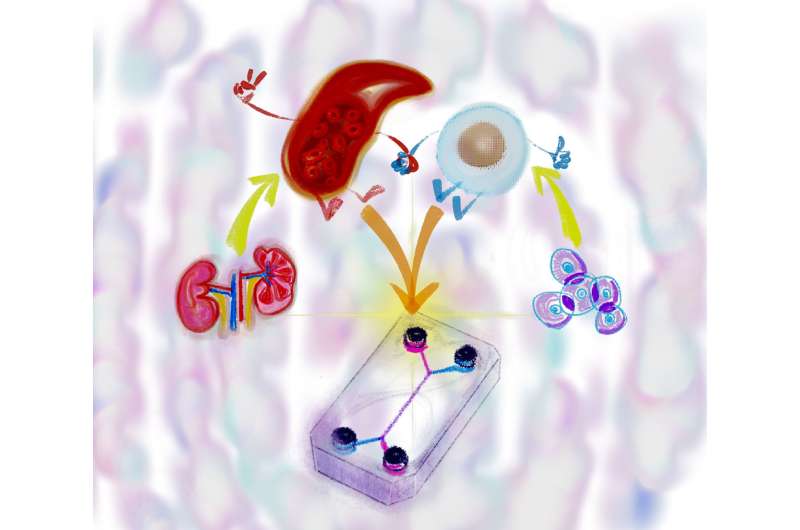
A team of researchers from Kyoto University has now developed a model microchip using human iPS cells to measure the transport capacity of the membrane proteins and potentially give test animals some respite. The paper, "Cells sorted off hiPSC-derived kidney organoids coupled with immortalized cells reliably model the proximal tubule," was published in Communications Biology.
"We focused on the fact that human iPSC-derived organoids contain highly functional cells whose functions are enhanced when cultured on a microfluidic device," explains lead author Ramin Banan Sadeghian of KyotoU's Graduate School of Engineering.
This microphysiological system—or MPS—is designed to reproduce the mechanisms of glucose reabsorption and drug excretion in the renal proximal tubules in vitro and ex vivo, mimicking human epithelial cells.
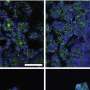
"We found that glucose uptake and transport are at significantly higher rates through the engineered co-culture tissue than through monocultures," says Toshikazu Araoka of KyotoU's Center for iPS Cell Research and Application, CiRA, referring to the team's use of a blend of pluripotent cell-derived and immortalized proximal tubule epithelial cells.
"Our results also showed that shear stress stimulation of the two cell types increased the transport capacities of the membrane proteins SGLT-2 and P-gp," adds team leader Minoru Takasato at RIKEN Center for Frontier Biosciences.
Takashi Yokokawa's team anticipates applying their MPS model as a screening tool for newly developed drugs by evaluating the transport and nephrotoxicity of various membrane proteins.
For example, these methods may enable patient-specific disease modeling, drug screening, and pathogenesis study by incorporating patient-derived stem cells into the required organoids.
"Our model will enable us to obtain the diseased cells that can be co-cultured with existing cell lines to form a homogenous tissue for analysis and treatment purposes," notes Yokokawa.
More information: Ramin Banan Sadeghian et al, Cells sorted off hiPSC-derived kidney organoids coupled with immortalized cells reliably model the proximal tubule, Communications Biology (2023). DOI: 10.1038/s42003-023-04862-7
Journal information: Communications Biology
Provided by Kyoto University