This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Using nanopore single-molecule sensing to identify glycans
Glycans perform varied and crucial functions in numerous cellular activities. The diverse roles of glycans are matched by their highly complex structures, which derive from differences in composition, branching, regio- and stereochemistry, and modification. This incomparable structural diversity is challenging to the structural analysis of glycans.
Recently, a joint research group led by Prof. Qing Guangyan and Prof. Liang Xinmiao from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) has developed a glycan identification method based on nanopore single-molecule sensing through a glycan derivatization strategy. The study was published in Nature Communications on March 28.
Identifying and sequencing glycans using nanopore single-molecule techniques has sparked interest; however, it has achieved little progress over the past dozen years. Only a handful of cases that focused on either high molecular weight polysaccharides or some monosaccharides were reported.
For smaller but structurally more diverse glycans with greater biological significance, single molecule detection with nanopores has not yet been achieved, largely because the fast passage of glycans through a nanopore cannot be sensed due to the small size and weak affinity of the glycan with the nanopore.
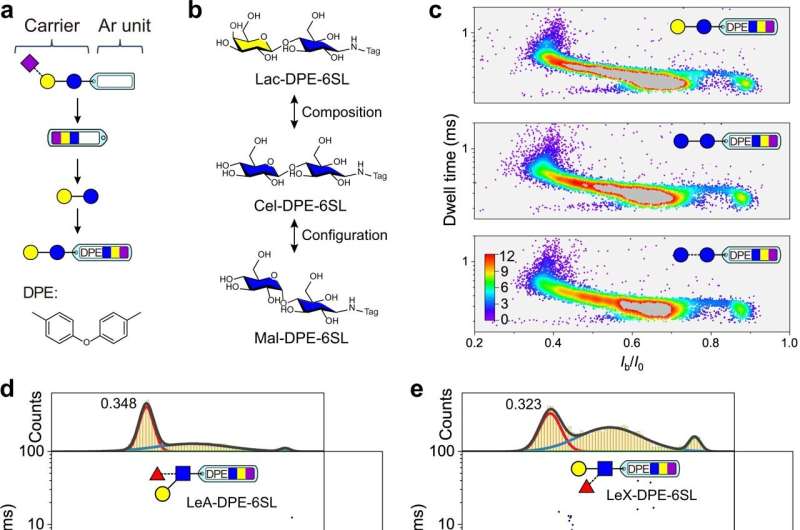
To address the challenge, the researchers introduced a derivatization strategy by linking an aromatic-type tag group to small glycans via a high-efficiency and facile reductive amination reaction. The resulting tagged glycan was sensed with a wild-type aerolysin nanopore by presenting strong nanopore blockage signals.
The researchers obtained a scatter plot based on blockage current and dwell time as the fingerprint map by processing the nanopore single-molecule blockage events. They identified different glycan isomers, glycans with varying lengths, and branched simple glycans.
Moreover, they revealed that multiple cation-π interactions between the aromatic tag of glycan with K238 residues of the nanopore interface retarded the translocation of the tagged glycan and contributed to sensing.
"This study pushes the boundary of nanopore sensing beyond its traditional focus on nucleic acid and protein, and activates its power in the glycomics and glycoscience field, which might pave the way towards nanopore glycan sequencing," said Prof. Qing.
More information: Minmin Li et al, Identification of tagged glycans with a protein nanopore, Nature Communications (2023). DOI: 10.1038/s41467-023-37348-5
Journal information: Nature Communications
Provided by Chinese Academy of Sciences