This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Ingestible capsule to address GI tract diseases
Diagnosing and treating gastrointestinal tract diseases can be notoriously invasive and time-consuming: blood and stool lab work; biopsies, colonoscopies and endoscopies; and X-rays, CT scans and MRI imaging. But what if there was an alternative as simple as popping a Bayer aspirin?
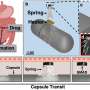
Researchers in the University of Maryland's MEMS Sensors and Actuators Laboratory (MSAL) in the A. James Clark School of Engineering have developed an ingestible capsule with a new packaging technology that can protect its tiny components in the sometimes harsh environment of the GI tract, then dissolve at precise moments and locations needed to deliver drugs, reveal sensors or carry out other functions.
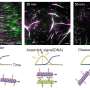
Their study, published in Microsystems & Nanoengineering, describes how this packaging, called a freestanding region-responsive bilayer (FRRB), can protect the capsule as it navigates the GI tract and performs complex diagnostic and therapeutic tasks like sensing, monitoring and drug delivery.
"Ingestible capsule devices are the next frontier of medical technology," said bioengineering Ph.D. student Michael Straker, first author of the paper. "The FRRB is a simple yet elegant solution to one of the major challenges of developing these devices. It can be used to develop creative new designs, allowing sensitive actuators and sensors to reach targeted regions of the GI tract unscathed."
Co-authors on the paper include materials science and engineering Ph.D. student Joshua Levy, electrical and computer engineering Ph.D. candidate Justin Stine, Vivian Borbash '22, research associate Luke Beardslee and Herbert Rabin Distinguished Chair in Engineering Reza Ghodssi, who directs the MSAL.
Their work focuses on addressing autoimmune disorders like inflammatory bowel disease, which includes Crohn's disease and ulcerative colitis and affects more than 3 million Americans, according to the Centers for Disease Control and Prevention. It also could eventually be useful for conditions involving gut-brain connections, such as depression.
For many years, pharmaceutical companies have relied on pH-responsive materials, commonly known as an "enteric" coating, to deliver drugs into the body; it is used in the daily aspirin that many people take for heart health. But enteric coatings aren't sophisticated enough to protect an ingestible capsule from the acidic fluids and solids it encounters as it journeys through the GI tract en route to the intended destination: the intestines. It has to remain intact so that the components, such as the sensor or the drug, can be uncovered and released at the right time.
One idea that has been tried is adding an opening mechanism to the capsule. However, such mechanisms can be bulky and add complexity and weight to the device. They also may require stimulus from beyond the capsule, through high-powered equipment.
The FRRB developed by MSAL, a research group focused on microelectromechanical systems (MEMS) with a long history of groundbreaking research into ingestible capsules, is a more versatile option. It can be readily applied to various ingestible capsule components, can be fabricated into a number of shapes, and can protect those fragile components within the GI tract until they reach their intended destination, then dissolve away so they can perform the functions for which they were designed, in sequential order.
"It is our expectation that this manufacturing approach will widen the design paradigm of developing minimally invasive micro/nano/bio devices and systems for health care monitoring, treatment and prevention applications," said Ghodssi.
More information: Michael A. Straker et al, Freestanding region-responsive bilayer for functional packaging of ingestible devices, Microsystems & Nanoengineering (2023). DOI: 10.1038/s41378-023-00536-w
Provided by University of Maryland