This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Microbes that 'eat together' may benefit from a shared immunological memory

Viruses are the most abundant and diverse biological entities on Earth, living in every type of habitat. In the ocean alone viruses are ten times more abundant than microbes.
Viruses replicate by infecting living organisms, from humans and animals to insects, and even microbes. While environmental viruses that infect microbes is not a recent finding, their prevalence, however, was not previously known. Researchers are only now beginning to understand the diversity of viruses and their impact and function in ecosystems.
A new study published in Nature Microbiology examines viruses that infect microbes in the deep sea and finds evidence that viruses interact with a far more diverse set of hosts than was previously thought. The study's findings could aid in better understanding of viruses and in engineering virus therapies.
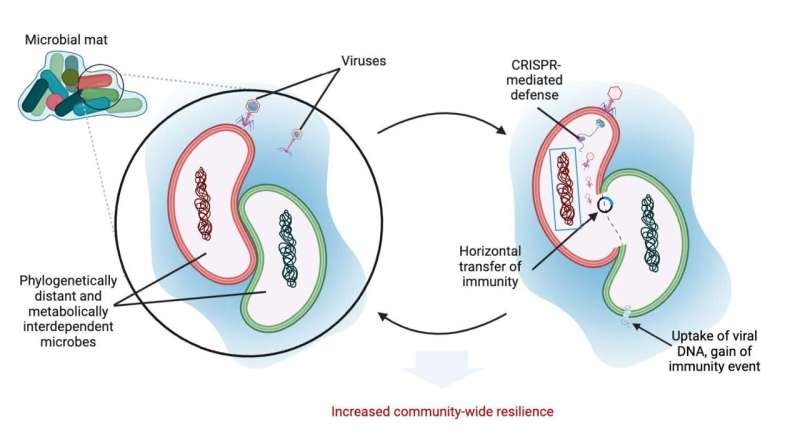
Lead author Ph.D. candidate Yunha Hwang and senior author Professor Peter Girguis, both in the Department of Organismic and Evolutionary Biology, collected samples from deep-sea hydrothermal vent microbial mats during a 2021 expedition in the Guaymas Basin, Mexico. These microbial mats are composed of massive numbers of bacteria and archaea, microbes that dominate in these ecosystems. Both are microbial, but bacteria and archaea are very distinct taxa; as different from each other as bacteria are from people.
Though worlds apart, many archaea and bacteria survive through symbiotic relationships. At hydrothermal vents, bacteria and archaea form masses that can harness energy from the methane found in these environments. While this relationship is necessary for survival, it does not change the fact that these two lineages are biologically very different. Which made it even more surprising to Hwang and Girguis to discover that both bacteria and archaea carried immunity against the same viruses.
"We were baffled when we saw the results," said Hwang, "because whether they're symbiotic or not, infection machinery is thought to be very complicated and host-specific. If archaea and bacteria are so different how can one virus infect both?" That question led the researchers to think about all the diverse ways in which viruses can interact with microbes that go beyond infection.
Most work on viruses has been done in laboratories with one culture and one virus. Studies have only recently begun to extend to natural environments, which requires the use of different tools as microbes are not easy to culture in labs.
"Around ninety-nine percent or more of the microbes that we know exists in nature we can't culture in the lab," Hwang said, "now we are able to sequence microbial DNA without culturing what's in the ocean or in the soil. And with that, we can ask, 'what are the viruses that are there and what are their interactions?'"
Before joining Girguis's lab, Hwang studied viruses in desert environments and observed that host-virus interactions in nature are much more nuanced than in laboratory settings. The deep-sea vents—in contrast to desert soils—harbor large microbial mats with billions of microbes engaging in symbiotic relations. In observing these unique environments the researchers asked, if microbes live in such high densities, are there viruses that have broader "host ranges?" In other words, were they capable of infecting diverse microbes?
They sequenced DNA from the samples and recovered metagenome-assembled genomes of the microbes and viruses. They used CRISPR spacers (which encode the microbe's immunological memory) to infer to which viruses in the sample the microbe is immune.
To confirm their findings, they employed a newer technique called Hi-C (high throughput chromosome conformation capture) sequencing. If viral DNA is found inside a cell, the Hi-C technique can sequence crosslinked viral and host DNA. Finding statistically significant contact between viral DNA and microbial DNA, the researchers could confirm their findings that viral DNA are in not just one type of cell, but phylogenetically distant cells as well.
"The CRISPR spacer analysis and Hi-C data showed a striking pattern that viruses genomically interact with very distantly related sets of microbes, particularly those that are in symbiosis with each other," said Hwang, "this interaction results in a very intriguing phenomenon where symbiotic microbes carry immunological memory against the same viruses, which means there is an advantage in symbiotic partners collaborating that exists also in their immunity. We have seen this within populations of bacteria, but we haven't seen it across distantly related species. This is quite a poignant finding in that it reveals how interconnected the natural environment is."
"Yunha is very clever to have designed an experiment that takes advantage of vent microbial mats to better understand the role of viruses in habitats where microbial densities are crazy high," said Girguis, "she's also very thoughtful in looking for patterns in the genomes of both archaea and bacteria. The CRISPR spacer and Hi-C data showed us that the bacteria interacted, in some way or another, with the same virus as the archaea, which is totally wild."
The study challenges the conventional wisdom that viruses interact with a narrow set of hosts. And while the researchers are still gathering the direct evidence of a single kind of virus infecting these two very different hosts, the data clearly shows evidence of both bacteria and archaea having immunity against the same virus.
These results led Hwang and Girguis to propose different models of host-virus interactions with ecological and evolutionary implications that go beyond infection. They suggest that viral interactions with microbes that are not the virus's primary hosts may actually be prevalent in nature, particularly where microbes exist in a symbiotic relationship.It's very unlikely the same virus can infect both bacteria and archaea," Hwang said. "Instead, we propose that either one partner gained and retained immunity after a non-infectious encounter with a virus, and/or immunity was horizontally transferred between symbiotic partners."
The polyvalent nature of host-virus interactions in natural environments, and the diverse modes of interaction beyond infection, present important considerations as researchers move towards using viruses for biotechnological and medical applications, such as virus therapy in natural environments like the gut.
"These host-virus interactions in natural environments show immunity can cross large phylogenetic distances resulting in inter-populations building greater viral resilience together," said Hwang.
More information: Yunha Hwang, Viruses interact with hosts that span distantly related microbial domains in dense hydrothermal mats, Nature Microbiology (2023). DOI: 10.1038/s41564-023-01347-5. www.nature.com/articles/s41564-023-01347-5
Journal information: Nature Microbiology
Provided by Harvard University