This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Researchers suggest metal labeling strategy for single-cell multiplexing with mass cytometry
Researchers from the Suzhou Institute of Biomedical Engineering and Technology (SIBET) of the Chinese Academy of Sciences (CAS) have recently proposed a novel metal labeling strategy to increase the sensitivity of mass cytometry (MC) and expand the number of MC detection channels.
MC is an emerging bioanalytical technique for the simultaneous interrogation of high-dimensional biomarkers on individual cells. By using heavy metal isotope-labeled antibodies (Abs) to stain cells, MC avoids the spectral overlap and auto-fluorescence phenomena that severely limit the highly multiplexed capabilities of conventional fluorescence flow cytometry (FCM) and theoretically allows the measurement and quantification of up to 135 markers with single-cell resolution.
However, more than 60% of the isotope channels have not been implemented due to the lack of suitable metal isotope trapping supports. Thus, a novel approach to synthesize MC mass tags is still full of challenges.
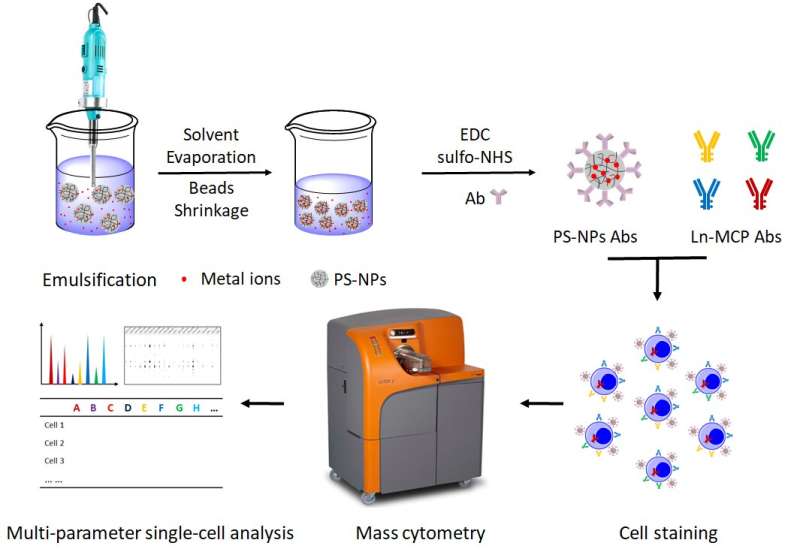
The strategy, proposed in this study by researchers led by Prof. Bai Pengli, is based on the using of a classical material, polystyrene nanoparticles (PS-NPs), to carry metals (rare earths, Zr, Hf) and conjugate with antibodies. The research was published in Journal of Colloid and Interface Science.
At present, several nanoparticle-based MC mass tags were reported to increase the sensitivity and expand MC detection channels. Nevertheless, most of them need complicated surface modifications during probe preparation in order to lower the nonspecific binding of mass tags to cells.
By using polystyrene nanoparticles as metal carriers, Liu Zhizhou and his colleagues in BAI's group proposed a universal approach to synthesize MC mass tags.
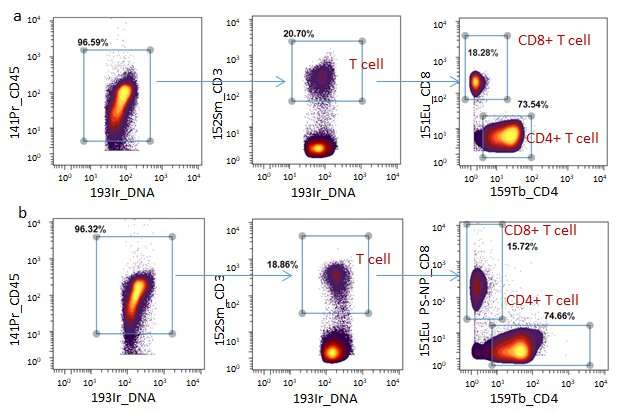
Four new MC detection channels (177Hf, 178Hf, 179Hf and 180Hf) have been introduced into MC for the first time. When used in conjunction with commercially available metal chelating polymer (MCP) mass tags, the metal@PS-NP_Abs mass tags can clearly distinguish different subpopulations of human mononuclear cells. The new mass tag showed five times higher sensitivity than MCP mass tags.
In addition, a new nonspecific binding reduction method was reported by exploring new cell staining buffer instead of complicated surface modification and strict size control used in other mass tag design strategy.
Moreover, these PS-NPs based mass tags showed high compatibility with commercial MCP-Abs for highly multiplexed detection of human MNCs biomarkers.
"We believe that more kinds of PS-NPs with different metals would be prepared for MC metal tags, and dual-functionality probes for both FCM and MC can be achieved if appropriate dyes were swelling to the PS-NPs along with metal ions, which can be used in both MC and flow cytometry or imaging," said Liu, first author of the study.
More information: Zhizhou Liu et al, A universal mass tag based on polystyrene nanoparticles for single-cell multiplexing with mass cytometry, Journal of Colloid and Interface Science (2023). DOI: 10.1016/j.jcis.2023.02.092
Provided by Chinese Academy of Sciences