This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New composite material to create green hydrogen
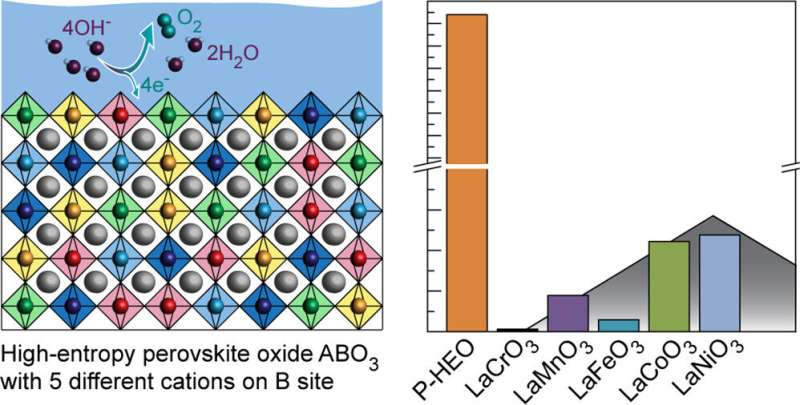
Researchers from the University of Twente developed a new composite material that outperforms the individual compounds by one to two orders of magnitude. The composite consists of several earth-abundant elements, that could potentially be used for efficient hydrogen generation without rare and precious metals like platinum. The researchers published their findings in the journal ACS Nano.
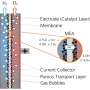
Green hydrogen is seen as the energy carrier of the future. Effectively, hydrogen offers a way to store (green) energy for long periods. This makes it especially important to produce it as efficiently as possible. Electrolysis of water is one of the most sustainable methods to produce green hydrogen. However, with current electrolysis methods, we need a lot of rare and expensive materials, or the process is not efficient enough.
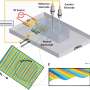
"Currently, the most efficient electrolyzers contain platinum and iridium, which are needed for the electrodes on which the hydrogen and oxygen gas are produced from water. However, platinum and especially iridium are too rare. That's why we're constantly looking for electrode materials made from more abundant resources which also can be used as efficient and stable electrocatalysts," explains UT-researcher Chris Baeumer. The researcher and his team found exactly what they were looking for in a new material, which is a compound containing five different transition metals.
Individually, the five transition metals are only moderately active when used as a catalyst. However, the researchers found that the combined activity outperforms the individual compounds by a factor up to 680. The higher activity comes as a surprise, explains Baeumer.
"We expected that the stability compared to traditional composites would be enhanced, but when we started testing it soon turned out that the activity was much higher too. In collaboration with our partners from Karlsruhe (Germany) and Berkeley (U.S.), we found that the individual transition metals may 'help' each other to make the combined material better than the sum of its parts in a so-called synergy-effect."
These new findings don't mean that we can directly replace all electrodes with this new material. Combining the five different materials is complex, and the activity was so far tested only in a lab-environment.
"We're comparing a newly discovered composite to materials optimized for large-scale production, meaning that our new material still needs to be tested on the industrial scale. However, with some tweaking and further research, this combination of transition metals has the potential to outperform currently available alternatives," explains UT-postdoc Shu Ni, who is leading these future developments for materials optimization.
More information: Mohana V. Kante et al, A High-Entropy Oxide as High-Activity Electrocatalyst for Water Oxidation, ACS Nano (2023). DOI: 10.1021/acsnano.2c08096
Journal information: ACS Nano
Provided by University of Twente