This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers engineer spinal cord-like tissue with drug-guiding function for spinal cord injury repair
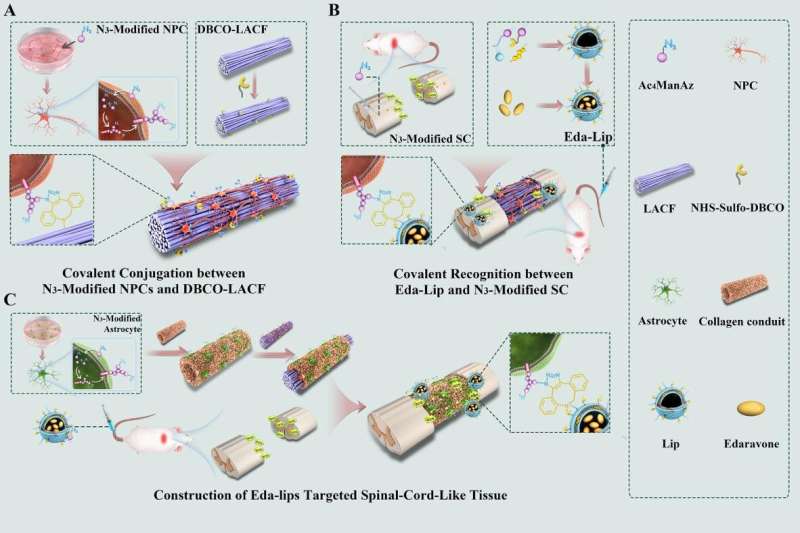
In a recent study published in Science Advances, a research team led by Profs. Dai Jianwu and Zhao Yannan at the Institute of Genetics and Developmental Biology (IGDB) of the Chinese Academy of Sciences demonstrated a strategy for covalent conjugation between biomaterials and cells to construct spinal cord-like tissue with drug-guiding function for spinal cord injury (SCI) repair.
SCI repair is one of the world's most challenging medical problems. Engineered spinal cord-like implants based on stem cells and biomaterials provide a new method for improving therapeutic outcomes for SCI. The adhesion of stem cells to scaffolds can be regulated by the stiffness, surface topological structure and porosity of the scaffold, as well as by the modification of integrin ligand or adhesion molecules based on noncovalent interactions.
However, the dynamic reversibility of noncovalent adhesion may be adverse for cell retention in complicated environments after SCI.
Unlike noncovalent interactions, covalent interactions form stronger bonds that hold atoms together by sharing electrons. However, whether covalent conjugation between biomaterials and stem cells can be used for tissue engineering has remained elusive.
In this study, the researchers first metabolically labeled human neural progenitor cells (NPCs) with Ac4ManNAz and modified the collagen scaffold NeuroRegen with dibenzocyclooctyne (DBCO) groups to create covalent interaction between biomaterials and cells.
Covalent conjugation significantly prolonged cell retention on the scaffolds, induced cells to spread along the direction of fibers and promoted cell differentiation, according to the researchers.
They then assembled the NeuroRegen scaffold containing NPCs into a collagen conduit containing N3-labeled human astrocytes to form an engineered spinal-cord-like transplant.
Based on the covalent recognition between N3-astrocytes and DBCO-modified nanoparticles, they delivered a neuroprotective agent, Edaravone, to attenuate oxidative stress after the implantation into SCI sites. They found that the implantation of an engineered spinal-cord-like transplant significantly promoted cell survival, neural regeneration, angiogenesis and locomotion function recovery in rats after SCI.
The researchers have been dedicated to SCI repair research for more than 20 years. The NeuroRegen scaffold they developed has been effective in clinical trials involving more than 100 patients. To further improve the therapeutic effect, they are carrying out a spinal cord tissue engineering project based on the NeuroRegen scaffold.
In order to select suitable seed cells for spinal cord tissue engineering, they have compared neural stem cells from different tissue origins and established a culture method for clinical-grade neural stem cells and astrocytes derived from human spinal cord tissue.
Through their other research and the present study, they are striving to construct human spinal-cord-like tissues that can be used for human SCI therapy.
More information: Weiyuan Liu et al, Spinal cord tissue engineering via covalent interaction between biomaterials and cells, Science Advances (2023). DOI: 10.1126/sciadv.ade8829
Journal information: Science Advances
Provided by Chinese Academy of Sciences