This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Single gene causes stinging cell to lose its sting
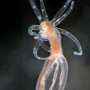
When scientists disabled a single regulatory gene in a species of sea anemone, a stinging cell that shoots a venomous miniature harpoon for hunting and self-defense shifted to shoot a sticky thread that entangles prey instead, according to a new study.
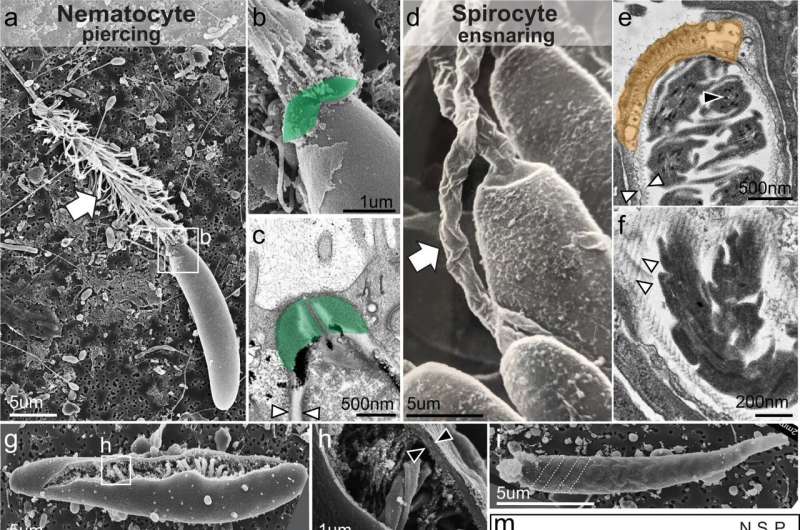
The research, carried out in the sea anemone Nematostella vectensis, shows how disabling a gene, called NvSox2, enabled a transition from a piercing cell (called a nematocyte), to a sticky, ensnaring cell (called a spirocyte). The finding suggests that the nematocyte cell may have evolved from a spirocyte, thanks to the development of the NvSox2 gene.
"This one gene controls a switch between two alternative cell fates; it controls a whole suite of traits that gave this cell a completely different identity," said Leslie Babonis, assistant professor of ecology and evolutionary biology at Cornell University.
Babonis is the corresponding author of "Single-Cell Atavism Reveals an Ancient Mechanism of Cell Type Diversification in a Sea Anemone" published in Nature Communications.
"Stinging cells" are found in all cnidarians—including sea anemones, corals, hydrae and jellyfish. They served as a model cell in the paper since they come in several dozen cell types, with different shapes and functions, allowing researchers to explore fundamental evolutionary questions of how a single cell type can become extremely diverse with many different forms.
At its core, this line of study seeks to better understand the evolution of animal diversity, as all life forms originated from single-celled organisms that became more complex as cells specialized and differentiated over time.
The findings underscore the fact that a kind of flexibility of function is built into the genetic architecture of stinging cells in N. vectensis. For example, if a small population of N. vectensis were to move into a new environment where a sticky thread proved more advantageous than a piercing harpoon cell, it would take only a small mutation in one gene to make the switch.
"Being able to 'choose' between different cell types gives an animal a lot of flexibility to invade new habitats and evolve new traits," Babonis said.
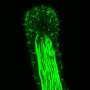
Nematocytes and spirocytes both contain a novel organelle composed of a thick, pressurized capsule. When prey or predator is detected nearby, the pressurized capsule collapses, forcing a projectile out of the cell—a harpoon in the case of nematocytes, and a sticky prey-entangling thread in spirocytes.
Babonis and colleagues used CRISPR/Cas9 gene editing to knock out an NvSox2, a transcription factor that binds to DNA and changes the expression of genes downstream. By doing so, the researchers discovered that NvSox2's role was to silence the development of sticky cells and promote the development of piercing cells in their place.
"The cells looked completely different and had a completely different function than the cells in the wild-type animals," Babonis said.
In future work, Babonis and colleagues plan to investigate the breadth of this phenomenon by searching for the same single-gene control over two cell fates in other species of cnidarians, including a closely related species of coral. A long term goal of the project is to work backward to identify the minimum set of genes needed to make a stinging cell that can still shoot a projectile. From there, they will experiment with variations.
"Can we make a type of stinging cell that has never evolved before?" Babonis asked. For example, she said, a tiny cell that shoots a small hypodermic needle could have valuable medical applications.
More information: Leslie S. Babonis et al, Single-cell atavism reveals an ancient mechanism of cell type diversification in a sea anemone, Nature Communications (2023). DOI: 10.1038/s41467-023-36615-9
Journal information: Nature Communications
Provided by Cornell University