This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
New nanotransporter for drug delivery inside cells

A new study by the University of Barcelona has analyzed the viability of a new nanomolecule as drug delivery vehicle. The results, published in the journal Colloids and Surfaces B: Biointerfaces, show that liposomes designed by researchers are able to transport and deliver an anticancer drug that has been used as a model inside cells.
The study included the participation of researchers from the UB faculties of Biology, Physics and Pharmacy and Food Sciences, as well as researchers from the Scientific and Technological Centers of the UB (CCiTUB), the Institute of Nanoscience and Nanotechnology of the UB (IN2UB) and from the Institute for Bioengineering of Catalonia (IBEC).
Interaction between liposomes and the cell membrane
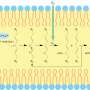
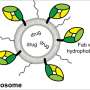
A liposome is an artificial spherical vesicle with a membrane made of a dual layer of lipids that is similar to the structure of cell membranes. Since these molecules were found in the 1960s, they have been used as models to study cell membranes and as potential drug delivery systems.
One of the challenges of turning liposomes into drug delivery vehicles is finding out how they interact with cell membranes and what basic mechanism—adsorption, fusion or endocytosis, or a combination of these three—is involved in the integration of liposomes by cells. "The interactions between liposomes and the cell membrane can be extremely different depending on the nature of the cell membrane and the lipidic composition of liposomes," the researchers note.
The liposomes designed by the UB team are small lipid spheres with a composition similar to that of the cell they want to treat. "This similarity eases its incorporation and the drug delivery inside the cell," notes Òscar Domenech, IN2UB member and one of the researchers that took part in the study.
Study in cell cultures
This study is the continuation of a previous study carried out by the same research team, which analyzed the fusion mechanisms of liposomes using a simplified model that copied the membrane of HeLa cells, a type of culture cells which are widely used in scientific research. "The membrane of HeLa cells is more complex than the model we used in the previous study. Now, we have used real cell cultures in order to obtain a better view of the interaction mechanism of our liposomes," the researchers say.
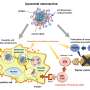
To study the interaction between the liposomes and the cell membrane and to assess the integration of these nanomolecules, the researchers combined two techniques. On the one hand, they used confocal fluorescence, which allows them to see fluorescent molecules inside the cell. Then, the liposomes encapsulated calcein, a fluorescent dye, to see whether the nanomolecule and its content were entering the cells.
On the other hand, the researchers used the atomic force microscopy technique to see the physicochemical changes of the cell surface and assess the stiffness of the cell membrane in the presence of liposomes. The interaction of the liposomes designed by the researchers affirmed the results obtained with model membranes and it shows the capacity of the formulation of these nanomolecules as a potential nanotransporter.
"We show that the lipid composition enables a delivery of the liposome content inside the cell, as well as the effect of the filopodia—small flagella of the cell—in easing the arrival of liposomes at the cell membrane," adds Òscar Domènech.
A test with an anticancer drug
To validate the capacity of these liposomes as a drug delivery system, the researchers encapsulated methotrexate, an immunosuppressive drug used in the treatment of several oncologic, inflammatory and autoimmune pathologies. "We could show that our liposomes are ideal for delivering this model molecule, which we know can eliminate cancer cells," notes Domènech.
These results open the door to future studies with other molecules and cell types. "Our interest would be to extend the methodology to other types of cells or even tissues to show the viability of the analysis, as well as to use other therapeutical molecules encapsulated in liposomes," says Domènech.
"Moreover, the two techniques we applied during the study allowed us to obtain results in a fast and minimally invasive way, which in the future could be indicators of the good functioning of a drug against cancer cells."
More information: Adrià Botet-Carreras et al, On the uptake of cationic liposomes by cells: From changes in elasticity to internalization, Colloids and Surfaces B: Biointerfaces (2022). DOI: 10.1016/j.colsurfb.2022.112968
Provided by University of Barcelona