This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New insights into a fundamental process in mitochondria
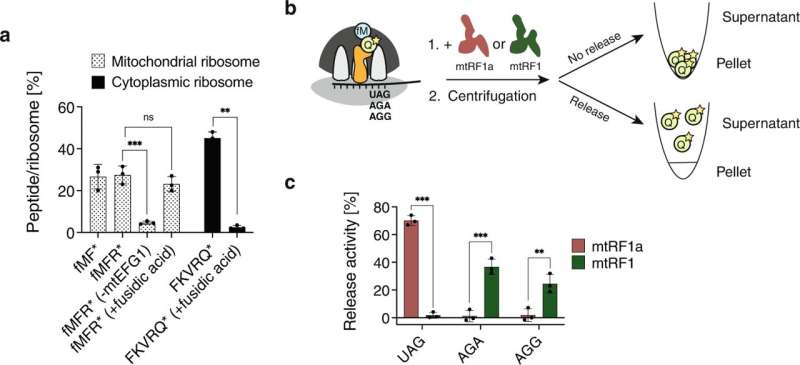
Researchers at Karolinska Institutet have uncovered the function of the release factor mtRF1 in the final steps of mitochondrial protein synthesis. The study, published in Nature Communications, reveals new insights into a fundamental process in mitochondria.
Mitochondria are specialized organelles within the eukaryotic cell. They originate from a bacterial ancestor and maintain their own DNA molecules and gene expression machineries. Due to their importance in cellular energy metabolism, they are often referred to as the "power-house of the cell."
The conversion of energy is carried out by large protein complexes located in the inner membrane of mitochondria. Central parts of these complexes are synthesized within the organelle. The synthesis pathway is unique, and many aspects of this process are still unresolved.
Combination of techniques
The researchers applied a combination of techniques, including ribosome profiling and a newly developed in vitro translation assay, to shine light on the final steps of protein synthesis in mitochondria.
"The mitochondrial genetic code differs slightly from the nuclear one. We show that two uncanonical codons, which's function has been unknown, are recognized by the release factor mtRF1, signaling termination of protein translation," says Annika Krüger, postdoctoral researcher at the Department of Medical Biochemistry and Biophysics at Karolinska Institutet.
"This finding highlights that while protein synthesis in bacteria and eukaryotic cytosol have been well described for decades, we are still discovering the basic components of analogical processes in mitochondria. There is still a lot to learn," notes Joanna Rorbach, senior author of the study.
More information: Annika Krüger et al, Human mitochondria require mtRF1 for translation termination at non-canonical stop codons, Nature Communications (2023). DOI: 10.1038/s41467-022-35684-6
Journal information: Nature Communications
Provided by Karolinska Institutet