The offshoot of cells visualized in real time
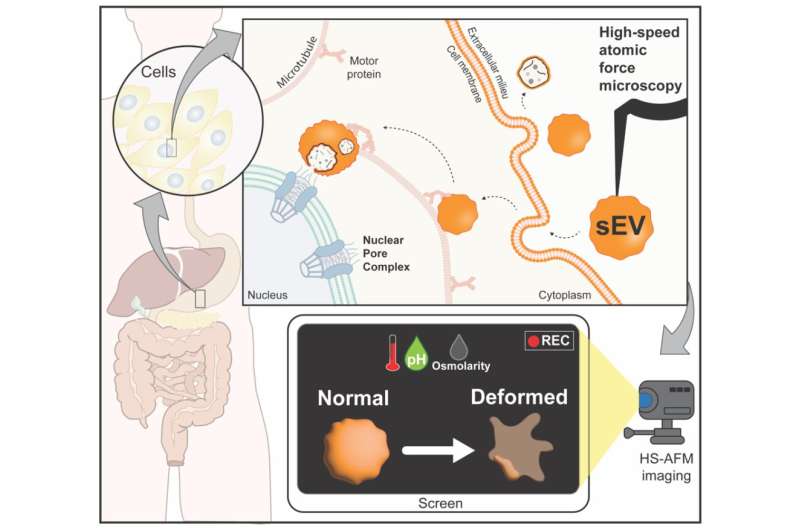
In a study recently published in the Journal of Extracellular Vesicles, researchers from Kanazawa University use high-speed microscopy to capture the dynamics of nanosized sacs released from cells.
Small extracellular vesicles (sEVs) are tiny sacs released by cells to deliver chemical messengers to other cells. Since sEVs are compatible with biological tissue they are being investigated as carriers for nanodrugs. However, the impact of physiological stress—such as changes in temperature—on the structure of sEVs is obscure. A research team led by Richard Wong and Keesiang Lim at Kanazawa University has now used an advanced form of microscopy to elucidate these changes in real time.
The temperature, acid, and salt levels in our bodies can fluctuate with factors such as disease. Thus, research on sEVs for drug development requires a deeper understanding of how stressful environments affect the vesicles' structure. For their study, the team first isolated sEVs from cells. Next, using a technique known as high-speed atomic force microscopy (HS-AFM) the structure of sEVs was revealed to be either spherical or ellipsoidal in shape. HS-AFM also enabled the researchers to accurately measure the sizes of sEVs without rupturing or damaging the vesicular membranes.
The effect of varying temperatures on sEVs was the first parameter assessed. At temperatures higher than normal (37°C) body temperature the vesicles showed deformations in shape coupled with a loss of elasticity of their membranes. On the other hand, sEVs in cold conditions (4°C) had a reduced ability to release any internal material effectively.
The researchers then studied the effects of pH (acid levels) on sEVs. The physiological pH of the bloodstream is 7.4. A pH less than 7 indicates acidic conditions and anything more than that is termed alkaline. The sEVs seemed to maintain their shape in acidic conditions (pH 4) but in alkaline conditions (pH 10) they were deformed. However, at a pH of 4 the sEVs were smaller in size suggesting their internal contents had been lost.
Now, salt levels (known as osmotic pressure) at a concentration of 0.15 M are healthy. However, changes in osmotic pressure can have detrimental effects on cells. As conditions were gradually changed it was found that the spherical nature of sEVs decreased at high salt concentrations (1.8 M) but seemed to remain intact at low concentrations (0 M). After a while, vesicles in high osmotic conditions showed ruptured membranes.
An understanding of these dynamics is imperative to formulating sEVs as pharmaceutical aids in different disease conditions. This study established HS-AFM as a useful tool to depict changes in sEVs under various physiological conditions in real time. "In summary, our study demonstrates the feasibility of HS-AFM for structural characterization and assessment of nanoparticles," concludes the team.
Small extracellular vesicles (sEVs) are sacs containing chemicals such as proteins, DNA, or metabolites released by cells. Usually playing the role of messengers, sEVs then deliver these chemicals to neighboring or distant cells. Hence, their shape, size, and elasticity are important for sEVs to function properly. For example, sEVs with reduced membrane elasticity might not be able to release their contents effectively.
sEVs are typically less than 200 nm in size and ferry around very small molecules. They are, therefore, under investigation for their potential role in delivering nanosized drugs/vaccines or chemicals used for diagnosis. The physical properties of sEVs in under varying physiological stress are, thus, important for understanding their behavior in different tissues and disease conditions.
Traditional AFM is a form of microscopy that utilizes a pointed lever grazing on the surface of molecules to depict their topography. As the lever moves about, it transmits signals that help create an image of the molecule's structure. However, the rate at which signals are emitted in AFM are too low to capture any dynamic changes. HS-AFM, on the other hand, enables a faster scanning of molecules such that images can be acquired in real-time. Even minor changes to a molecule's structure can be accurately captured.
More information: Elma Sakinatus Sajidah et al, Spatiotemporal tracking of small extracellular vesicle nanotopology in response to physicochemical stresses revealed by HS‐AFM, Journal of Extracellular Vesicles (2022). DOI: 10.1002/jev2.12275
Provided by Kanazawa University