Describing the mechanism by which oxygen in the blood is detected
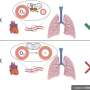
Hypoxia (oxygen deficiency), in addition to occurring in areas of considerable altitude, is a critical factor linked to various cardiopulmonary diseases of high morbidity and mortality in humans. The carotid body, a highly irrigated structure located in the carotid bifurcation, is considered the prototypical organ for the acute detection of oxygen.
Its activation during hypoxia causes hyperventilation and other cardiovascular reflexes, responses that are essential for adaptation to a reduced oxygen supply and that minimize its deleterious effects. Despite its biomedical relevance, the molecular basis for the acute detection of hypoxia has remained elusive for decades.
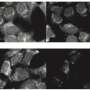
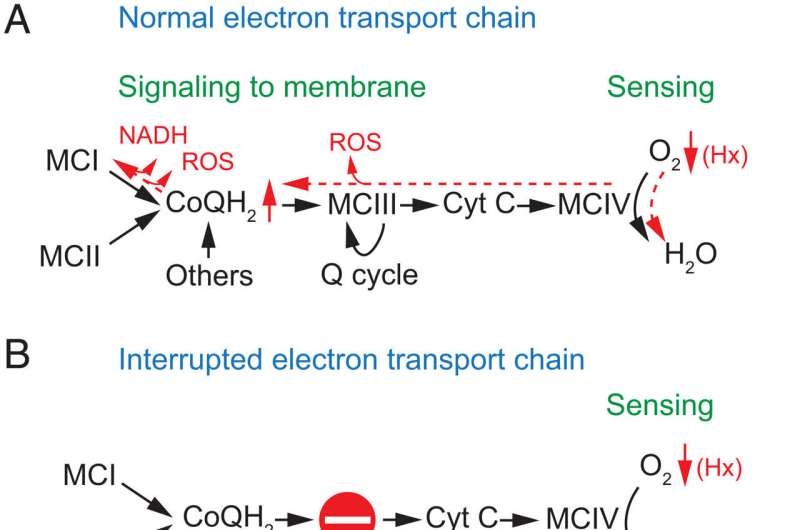
In previous studies, the group led by University of Seville professor José López Barneo showed that chemoreceptor cells of the carotid body (glomus cells) contain specialized mitochondria that during hypoxia create signals (including reactive oxygen species [ROS]) that regulate cellular excitability.
This specific sensitivity of the mitochondria of glomus cells to hypoxia is due to their specialized metabolism and depends on transcription factors, enzymes and specific components of the mitochondrial electron transport chain. The study, now published in the journal Proceedings of the National Academy of Sciences, completes the characterization of the molecular mechanisms for the acute detection of oxygen by the carotid body, a process with potential pathophysiological relevance in the poor adaptation responses to hypoxia.
Genetically modified mouse
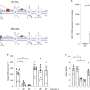
To this end, the researchers used as a model a genetically modified mouse in which the electron transport chain is interrupted in the carotid body. In particular, the studies were based on a mouse knockout for mitochondrial complex III of the respiratory chain in glomus cells, which results in the functional disconnection between mitochondrial complexes I and IV.
The chemoreceptor cells of the carotid body survive the dysfunction of mitochondrial complex III but show selective abolition of the cellular response to hypoxia, while the responses to other stimuli such as hypoglycemia remain. Accordingly, the mice present strong inhibition of the hypoxic ventilatory response, with a respiratory rate insufficient to make up for the oxygen deficiency.
This poor adaptation was revealed when the mitochondrial III complex-deficient mice were subjected to sustained hypoxia for days, given that the animals showed symptoms of abnormal acclimatization (excessive increase in hematocrit and cardiac hypertrophy, among others). The results indicate that, for an appropriate acute detection of hypoxia in the cells of the carotid body, a functional electron transport chain is required, where the integrated action of its components makes respiration regulation by oxygen possible.
Given the importance of the carotid body in regulating respiration, the researchers highlight that the mitochondrial electron transport chain emerges as a potential therapeutic target for drug treatment of respiratory depression and diseases in which overactivation of the carotid body is involved.
More information: Daniel Cabello-Rivera et al, Oxygen regulation of breathing is abolished in mitochondrial complex III-deficient arterial chemoreceptors, Proceedings of the National Academy of Sciences (2022). DOI: 10.1073/pnas.2202178119
Journal information: Proceedings of the National Academy of Sciences
Provided by University of Seville