Using spectroscopy technology, researchers discover cascade nanozymes can treat tumor cells
The equilibrium between reducing species and oxidizing species within the tumor microenvironment (TME) plays a crucial role in a majority of biological processes, especially the apoptotic cell death process. When the balance of oxidizing and reducing molecules is broken by the increase of reactive oxygen species (ROS) concentration, the cancer cells will die, achieving the purpose of tumor treatment.
At present, some new chemodynamic therapy and photodynamic therapy (PDT) methods based on nanozyme catalysis are used in the field of tumor therapy, aiming to achieve the effect of in-situ catalytic production of ROS in tumors. However, most of the studies on the mechanism of such treatments still only focus on the results of the cascade catalytic reaction of the nanozyme and cannot monitor the whole treatment process.
Surface enhanced Raman spectroscopy (SERS) is a rapid and non-destructive detection technology at the level of single molecule detection, which has great potential in monitoring related biochemical reactions in cells. The application of SERS technology to monitor the photodynamic therapy process of tumors furthers our understanding of the specific mechanism of the catalytic process of nanozymes, and can uncover specific information on the redox state in the tumor microenvironment.
In a new paper published in Light: Science & Applications, a team of scientists, led by Prof. Wei Song, Prof. Zhuo Liu and Prof. Bing Zhao from Jilin University, China, reported that cascade nanozymes of Au@CDs could be used for the photodynamic therapy of tumor cells. The SERS technique has been used to identify the dynamic change of ROS in TME throughout the whole PDT process. Spectroscopy is the best tool to monitor the intracellular biochemical reactions with a superior sensitivity. This work presents an insightful study about the PDT mechanism based on oxidative stress injury in TME, providing a powerful means for the real-time monitoring of tumor phototherapy.
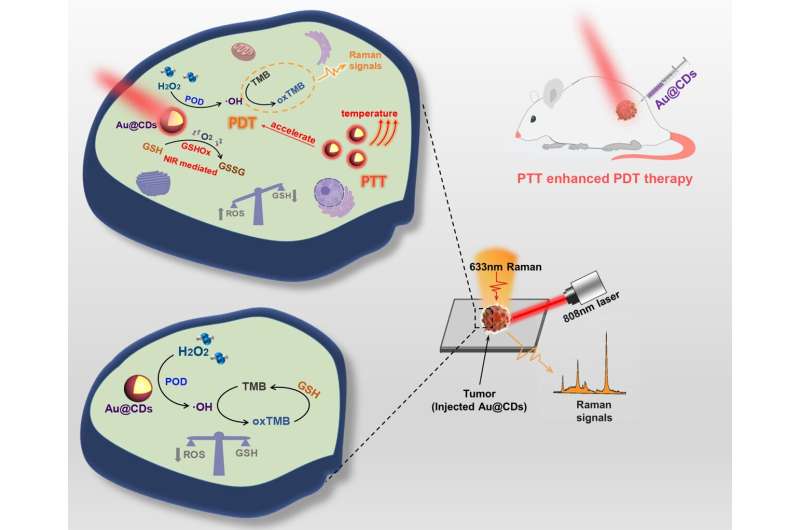
The researchers designed an Au@CDs cascade nanozyme, in which the CDs are used as a template and capping agent to construct typical core-shell structure Au@CDs nanoparticles (NPs). Compared with the Au NPs alone, the CDs shell avoids the aggregation of Au nuclei and provides a dense and uniform SERS hotspot. Under the excitation of near-infrared (NIR) light at 808 nm, Au@CDs exhibit NIR light-induced enhanced peroxidase like (POD) and NIR light induced glutathione like oxidase (GSHOX) activity.
Under the irradiation of NIR light, a large number of hot carriers excited by surface plasmon resonance (SPR) can effectively participate in the reaction, and the typical plasma photothermal effect of Au NPs can enhance the POD activity. In addition, Au@CDs mediate glutathione (GSH) to participate in the reaction, which accelerates the generation of ROS and presents the photodynamic treatment effect of photothermal enhancement. This cascade of nanozyme catalytic processes will rapidly break the redox homeostasis in tumors, generate a large amount of ROS, and eventually lead to cancer cell apoptosis (Figure 1).
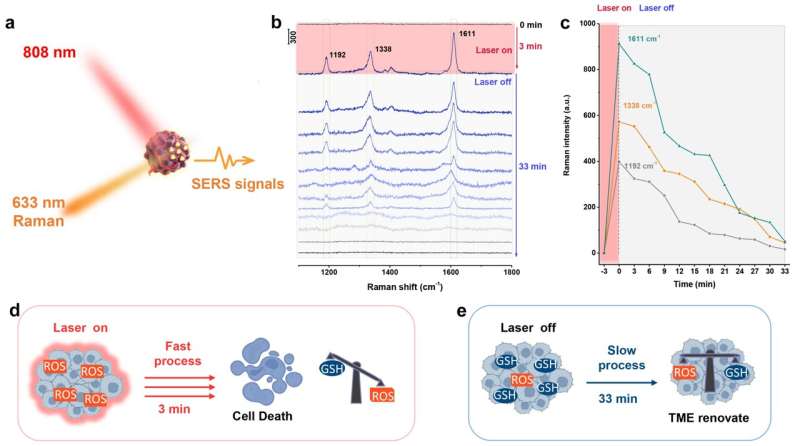
SERS technology is used to monitor the dynamic change of ROS in the tumor microenvironment during photodynamic therapy by recognizing the oxidation products of 3,3,'5,5'-tetramethylbenzidine (TMB) substrate molecules. Specifically, the enhanced effect of photothermal property under NIR laser irradiation for PDT processes can catalyze the decomposition of H2O2 and the decrease of GSH level in a very short time, further amplifying the reactive oxygen species damage that leads to tumor elimination.
Subsequently, the signals of oxTMB decrease over 33 min upon turning off the NIR laser, which is very slow compared to the increase process mentioned above under laser irradiation (3 min) (Figure 2). This result further suggests that overexpressed reductants (such as GSH) in TME will consume over-expressed ROS again, then until the ROS activity is counteracted and eliminated, the oxidation-reduction equilibrium state will be realized again.
Furthermore, through SERS strategies, a complete oxidative stress process in TMB is revealed. This insightful study about the reaction mechanisms provides the most valuable data support for the real-time monitoring of tumor phototherapy and normal tissue self-repairing.
More information: Linjia Li et al, SERS monitoring of photoinduced-enhanced oxidative stress amplifier on Au@carbon dots for tumor catalytic therapy, Light: Science & Applications (2022). DOI: 10.1038/s41377-022-00968-5
Journal information: Light: Science & Applications
Provided by Chinese Academy of Sciences