October 28, 2022 report
Synthesizing quantum nanomagnets via metal-free multi-porphyrin systems
A team of researchers at Shanghai Jiao Tong University, working with a pair of colleagues from Harvard University, has developed a new way to synthesize single quantum nanomagnets that are based on metal-free, multi-porphyrin systems. In their paper published in the journal Nature Chemistry, the group describes their method and possible uses for it.
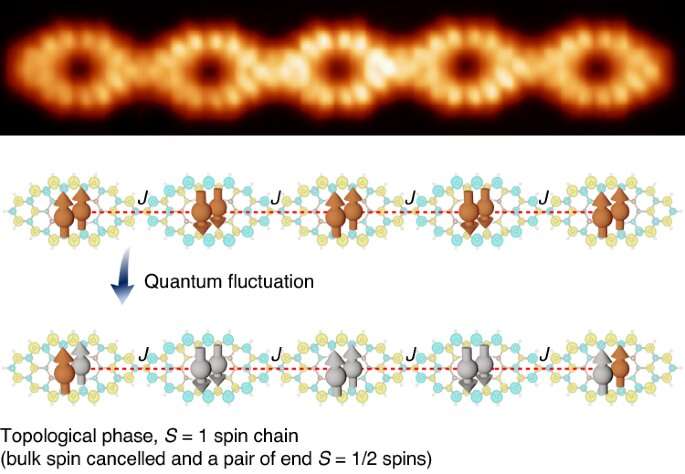
Molecular magnets are materials that are capable of exhibiting ferromagnetism. They are different from other magnets because their building blocks are composed of organic molecules or a combination of coordination compounds. Chemists have been studying their properties with the goal of using them to develop medical therapies such advanced magnetic resonance imaging, new kinds of chemotherapy and possibly magnetic-field-induced local hyperthermia therapy. In this new effort, the researchers have developed a way to create molecular nanomagnets with quantum properties.
The technique involved first synthesizing a monoporphyrin using what they describe as conventional "solution chemistry"—the monoporhyrins were created by using an atomic-force microscope to pull hydrogen atoms off of polyporphyrins. The researchers then applied the result to a base of gold, which they placed in an oven and heated to 80 °C. This forced the rings in the material to become chained. They then turned the oven up to 290°C and then let the material cook for another 10 minutes. This resulted in the formation of additional carbon cycles and the creation of quantum nanomagnets.
The technique works because it involves the use of porphyrins, which are heterocyclic molecules that have multiple double-bonds with delocalized electrons. They typically exist as rings. They also easily form complexes with ions and rare earth metals, which allows them to be used to create molecular magnets.
Once their magnets were complete, the researchers studied them using a scanning-tunneling microscope, finding ferromagnetic interactions between spins of 15 millielectronvolts. They further confirmed the existence of magnetic interaction exchange using spectroscopy with spin circulation.
The researchers suggest their approach is a relatively easy way to make polyporphyrin quantum nanomagnets of variable lengths, which can also have differing numbers of radical centers.
More information: Yan Zhao et al, Quantum nanomagnets in on-surface metal-free porphyrin chains, Nature Chemistry (2022). DOI: 10.1038/s41557-022-01061-5
Journal information: Nature Chemistry
© 2022 Science X Network