Better metal oxides to boost the green credentials of many energy applications
Researchers have solved a key hurdle in greener manufacturing, carbon capture, energy storage and gas purification—using metal oxides.
Metal oxides are compounds that play a crucial role in processes that reduce carbon dioxide (CO2) emissions. These processes include carbon capture, utilization and storage (CCUS), purifying and recycling inert gases in solar panel manufacturing, thermochemical energy storage, and producing hydrogen for energy.
These processes are based on reactions where metal oxides gain and lose electrons, known as redox reactions. However, the performance of metal oxides suffers under redox reactions at the high temperatures required for chemical manufacturing.
Now, a team led by Imperial College London has developed a new materials design strategy that produces copper-based metal oxides that perform better under high temperatures. The technology is already having a global impact on argon recycling in solar panel manufacturing and is expected to help unleash even more power from existing energy technologies that fight the climate crisis.
Senior author Dr. Qilei Song, of Imperial's Department of Chemical Engineering, says that "as the world transitions to net zero, we need more innovative industrial processes for decarbonization. To enhance energy security, we must diversify the electricity supply, from renewable energy generation and storage to clean use of fossil fuels with CCUS technologies. Our improved metal oxides hold great potential for use in the energy processes that are helping us reach net zero."
The paper is published in Nature Communications.
Unpicking a process
Metal oxides are key players in a relatively new process called chemical looping combustion (CLC).
CLC is an alternative way of burning fossil fuels that uses metal oxides, such as copper oxides, to transport oxygen from the air to react with the fuel. The reaction produces CO2 and steam, which is condensed to allow the efficient capture of CO2 to prevent it entering the atmosphere
By capturing the CO2 that is produced, CLC can help people to use fossil fuels in a cleaner way, and is already used in the EU, U.S., and China.
However, a key issue that has held back CLC from use on a larger scale is metal oxides' inability to maintain good oxygen-releasing performance over multiple redox cycles at high temperatures.
To solve the problem, the researchers examined the fundamental structures of the metal oxides used in CLC, reasoning that the precursor chemistry to metal oxides was poorly understood, which limited their rational design.
Co-lead author Michael High, Ph.D. candidate at Imperial's Department of Chemical Engineering, says that "to solve the question of how metal oxides maintain their performance, we looked to the basics of the chemical processes involved in CLC. This is a key example of combining fundamental research and smart design to produce a strategy that's applicable to a wide range of engineering processes."
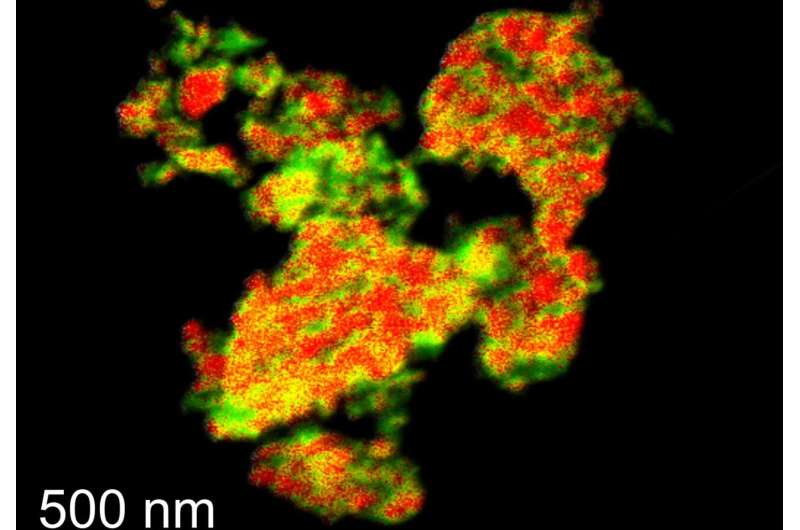
They used an alternative way to engineer the metal oxide structure from a well-known precursor composed of copper-magnesium-aluminum layered double hydroxides (LDHs). By tailoring the chemistry of LDH precursors, researchers found they could produce metal oxides that could still perform well under remarkably high temperatures. They demonstrated this by putting the oxides through 100 chemical cycles in a widely used type of reactor, known as a fluidized bed reactor, for 65 hours.
Their greater ability to withstand heat means that metal oxides produced in this way can be used to unleash more power from purifying and recycling inert gases like argon in manufacturing solar panels, capturing and storing carbon, chemical energy storage, and producing clean hydrogen. To show this, the researchers scaled up the production of metal oxides for use in fluidized bed reactors. They found that creating these materials is simple and readily suitable for upscaling using existing industrial manufacturing methods.
Senior author Professor Paul Fennell, also of the Department of Chemical Engineering, says that "the world must reach net zero carbon emissions by 2050. Renewable energies are developing rapidly, but in the short term we need to develop cost-effective carbon capture technologies that can be applied to decarbonize the industry. Our work will help solve this global challenge."
Next, the researchers will study the long-term stability of the materials during the combustion of different types of fuels, explore new applications for thermochemical energy storage, and extend the approach to other metal oxide systems for producing clean hydrogen via thermochemical redox cycles.
More information: Michael High et al, Precursor engineering of hydrotalcite-derived redox sorbents for reversible and stable thermochemical oxygen storage, Nature Communications (2022). DOI: 10.1038/s41467-022-32593-6
Journal information: Nature Communications
Provided by Imperial College London