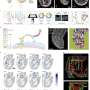
Single cell research spotlights the role of DNA methylation in cell fate decisions
Research applying single-cell analysis techniques and comparisons to a cell atlas resource allowed researchers at the Babraham Institute to connect observed development defects caused by disrupted DNA methylation processes with an understanding of the cell types affected. This work, published in Genome Biology, builds on previous work undertaken with collaborators to establish a detailed cell atlas charting cell fate through early development. This work contributes important new knowledge to understanding the role of DNA methylation during embryogenesis, helping decipher the rules that govern how different cell types arise. An understanding of these rules will be essential for researchers to be able to accurately and safely direct cell fate to produce clinically-relevant cell types for regenerative medicine.
Research from the Reik labat the Babraham Institute has advanced our understanding of the role of DNA methylation during the earlier stages of development. Technological advances in this field, providing the ability to collect parallel data types from a single cell and the existence of cell atlas references and comprehensive data sets, are revolutionizing what we know about the processes that determine cells fate. Elucidating the rules of how different cell types are formed has applications in regenerative medicine as well as in understanding developmental disorders and disease.
The erasure and reintroduction of DNA methylation is known to be vital in establishing cell identity as the tissues and organs of the embryo are formed. Deletion of key methylation enzymes in mice causes severe developmental defects and embryo lethality in some cases. Despite the importance of DNA methylation in development, the underlying mechanisms of how this is achieved is poorly understood. This is due to limitations on the information researchers could previously collect to understand the effects of changes in the usual processes of DNA methylation in development, which was restricted to the analysis of developmental defects, sample imaging and limited genome-wide analysis using bulked samples. These methods were not sufficient to resolve effects at the level of different cell types.
Using mice where key methylation enzymes were deleted, researchers from the Reik lab in the Institute's Epigenetics program performed single-cell gene expression analysis at the start of organ development, which occurs at day 8.5 after fertilization. Utilizing the power of single-cell approaches the researchers were able to follow which cell types were affected, in terms of not being able to form in the mouse embryo, suggesting the mechanisms behind the effects seen at a whole-organism scale.
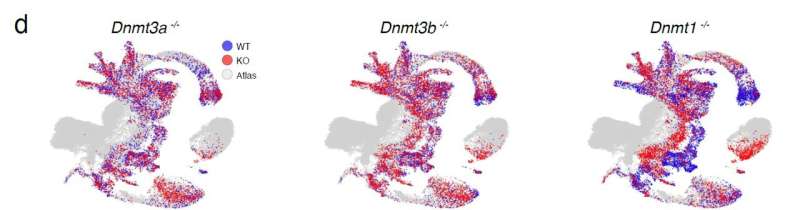
The research used genetically modified mice where two key groups of methylation enzymes were deleted: knock-out mice lines where DNA methyltransferases (DNMT 1, 3a and 3b) which introduce and maintain DNA methylation were individually deleted, and a system to investigate the effects of a combined deletion of all three TET enzymes (ten-eleven translocation (TET) methylcytosine dioxygenases) 1/2/3), which cause demethylation.
Dr. Stephen Clark, a senior researcher in the Reik lab when this research was undertaken, said, "The use of single-cell approaches is really providing the resolution we need to study the mechanics of DNA methylation during development. The picture we were able to build up confirms the repressive role of DNA methylation at this developmental timepoint, firstly that maintaining correct DNA methylation is required to suppress past and alternative cell type identities, and secondly that DNA methylation needs to be removed from parts of the genome to allow certain cell types to form."
A technique called single-cell RNA sequencing was used to measure gene expression across the genome in each mouse line. Comparing these expression profiles with a reference dataset allowed all the cell types of the embryo to be identified. Following that step, the effect of methylation perturbations on cell fate could be assessed by comparing the make-up of the knock-out embryos (where methylation enzymes were deleted) to wild type embryos at the same stage of development to highlight differences in cell type proportions.
The researchers were able to correlate effects on cell type formation at day 8.5 of development that matched observed phenotypes and analyze cell-type-specific changes in gene expression that could be linked to defects in cell fate commitment.
Dr. Ricard Argelaguet, a former postdoctoral researcher in the Reik lab at the Institute and co-first author on the paper, said, "The ability to both have the whole-organism perspective and the granularity of observing changes in cell types and gene expression has provided us with the ability to tease apart the role of DNA methylation and demethylation in the developing embryo at this particular timepoint to create new insights. It will be equally interesting to apply this approach to later timepoints to understand more about the role of DNA methylation as development progresses."
The research has created an interactive data platform providing gene expression readouts at the single-cell level from Dnmt and Tet mutant mice embryos.
Professor Wolf Reik, Director of the Altos Cambridge Institute of Science, who led the research while a group leader in the Epigenetics program at the Babraham Institute, said, "This research provides a rich resource to probe the connection between DNA methylation and the establishment of cell fate. This research benefited from published data sets and reference atlases and we hope that in turn, our work is of use to other researchers in both the development and epigenetics fields."
More information: Stephen J. Clark et al, Single-cell multi-omics profiling links dynamic DNA methylation to cell fate decisions during mouse early organogenesis, Genome Biology (2022). DOI: 10.1186/s13059-022-02762-3
Journal information: Genome Biology
Provided by Babraham Institute