Newts unleashed: Limb muscle regeneration needs metamorphosis and body growth

Unknown to passersby, a modest little creature with amazing abilities lives and breeds in the forests and paddy fields of Japan. Now, researchers from Japan have discovered how these amphibians' superpowers are unleashed.
In a study published this month in Scientific Reports, researchers from the University of Tsukuba have revealed that during limb regeneration in newts, two developmental processes—metamorphosis and body growth—are needed to provide the right conditions for muscle cells to be redeployed within the limb stump.
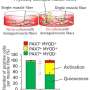
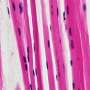
Newts, which are a semiaquatic type of salamander, are like most other amphibians in that they undergo metamorphosis. But unlike their relatives, newts are capable of repeated limb regeneration—even in the adult stage after they have undergone metamorphosis. In some newt species, individuals that have already metamorphosed regenerate muscle via dedifferentiation or reprogramming of muscle fibers in the limb stump, and mobilization of these fibers, to create muscle in the regenerating limb.
"Unlike cell differentiation, where cells become more specialized, cell dedifferentiation is a process via which they become less specialized," says senior author of the study, Professor Chikafumi Chiba. "Prior to our study, it was unknown whether metamorphosis or body growth was the key developmental process for muscle dedifferentiation."
The researchers investigated muscle cell dedifferentiation in the Japanese Fire-bellied Newt, Cynops pyrrhogaster, by tracking muscle fibers during limb regeneration while body growth and metamorphosis were experimentally delayed or advanced. The results suggest that metamorphosis and body growth are both needed for muscle differentiation.
Conversely, when larval newt muscles were cultured with a physiologically active thyroid hormone, tracking of the muscle fibers showed that these fibers can dedifferentiate independently of body growth and metamorphosis. These results indicate that newt muscle fibers have an inherent capacity to dedifferentiate, but that both body growth and metamorphosis are required for the fibers to activate this secret ability.
"We suggest that the developmental changes in the extracellular environment, or niche, inhibit the activity of myogenic stem cells—cells that can differentiate into muscle fibers—and promote the latent ability of muscle fibers to dedifferentiate. This way, the stem cells are compensated for by dedifferentiation, allowing newts to regenerate limb muscles throughout their life cycle," says Professor Chiba.
The results of this study provide an important foundation for future research on extracellular environments as well as the molecular mechanisms of dedifferentiation, such as the gene regulation that underpins this phenomenon. This research will also contribute to a deeper understanding of regeneration, and possible even to potential future medical treatments such as new therapies for diseases and muscle damage.
More information: Zhan Yang Yu et al, The latent dedifferentiation capacity of newt limb muscles is unleashed by a combination of metamorphosis and body growth, Scientific Reports (2022). DOI: 10.1038/s41598-022-15879-z
Journal information: Scientific Reports
Provided by University of Tsukuba