The black box behind embryonic development
DNA regulation is a critical process in a cell that allows it to fulfill its function. This process is key during pregnancy, when embryonic cells must develop into all cell types needed to form an embryo. An international team of researchers from KU Leuven, Babraham Institute, Radboud University, Ghent University and IMBA, have discovered that the first cell fate decision of embryonic development is regulated by a protein known as PRC2. This new insight into human development could help us to understand early pregnancy loss in the future. The results were published in Nature Cell Biology.
All human beings start their existence as a sperm cell and egg cell. By the fifth or sixth day, the fertilized egg has become a blastocyst, a rapidly dividing cluster of cells, consisting of an inner and outer group of cells. The outer group will form the placenta; the inner cells will develop into an embryo. This development is a complex process whereby every step is critical, and the slightest mistake may end embryonic development.
It's all about the right switch
Our bodies contain hundreds of different cells that perform a wide variety of functions and are part of the different tissues and organs. Interestingly, each cell in our body contains the same DNA. You might compare DNA to the fuse box in your house where—depending on where you need power—you switch the fuses on or off. The same happens in our cells: depending on the function of a cell, parts of the DNA are 'switched' on or off to activate the correct genes at the right time.
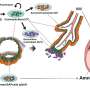
As early embryonic cells eventually form all cells, researchers have always assumed that early embryonic cells could easily switch genes on and off during development. "The first decision that cells need to take during human embryonic development is if they will form the embryo itself or the placenta. Our results show that this first decision is not as easy as we first thought," explains Professor Vincent Pasque (KU Leuven). "To form the placenta, the outer cells of the blastocyst must turn the correct switches on at the right time. We discovered that cells need to overcome barriers to turn placenta genes on," adds Hendrik Marks, group leader from Radboud University.
Understanding embryo development
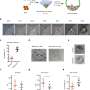
To study how the human embryo grows during the first week of development, the team has used blastoids, stem cell embryo models where stem cells organize into embryo-like structures and mimic embryo development. "By using this blastoid model, we could study human embryonic development," explains Nicolas Rivron, group leader at IMBA. "Blastoids recapitulate to some extent how human embryos behave during the first days of development."
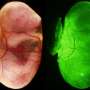
"Using this embryo model, we could see that when we remove the protein PRC2, an increased number of placenta cells are present in blastoids. These results show that PRC2 is a barrier for the placenta cells to appear," explains Ph.D. researcher Irene Talon (KU Leuven).
"Our research opens up the ability to better control stem cell specialization and blastoid development, which is useful for studying in the laboratory how the initial placental cells are formed. In the longer term, the new knowledge might improve our understanding of how embryos successfully implant in the uterus at the earliest stages of pregnancy, and why this can sometimes go wrong," concludes Professor Peter Rugg-Gunn (Babraham Institute).
More information: Maarten Dhaenens, Integrated multi-omics reveal polycomb repressive complex 2 restricts human trophoblast induction, Nature Cell Biology (2022). DOI: 10.1038/s41556-022-00932-w. www.nature.com/articles/s41556-022-00932-w
Journal information: Nature Cell Biology
Provided by KU Leuven