Carbon-coated nickel enables a hydrogen fuel cell free of precious metals
A nitrogen-doped, carbon-coated nickel anode can catalyze an essential reaction in hydrogen fuel cells at a fraction of the cost of the precious metals currently used, Cornell University researchers have found.
The new discovery could accelerate the widespread use of hydrogen fuel cells, which hold great promise as efficient, clean energy sources for vehicles and other applications.
It's one of a string of discoveries for the Héctor D. Abruña lab in their ongoing search for active, inexpensive, durable catalysts for use in alkaline fuel cells.
"This finding makes progress toward using efficient, clean hydrogen fuel cells in place of fossil fuels," said Abruña, the Emile M. Chamot Professor in the Department of Chemistry and Chemical Biology in the College of Arts and Sciences.
The results published March 21 in "A Completely Precious-Metal-Free Alkaline Fuel Cell With Enhanced Performance Using a Carbon-Coated Nickel Anode," in the Proceedings of the National Academy of Sciences.
Expensive precious metals, such as platinum, are currently required in hydrogen fuel cells to efficiently catalyze the reactions they employ to produce electricity. Although alkaline polymer electrolyte membrane fuel cells (APEMFCs) enable the use of non-precious metal electrocatalysts, they lack the necessary performance and durability to replace precious metal-based systems.
A fuel cell produces electricity through the hydrogen oxidation reaction (HOR) and an oxygen reduction reaction (OOR). Platinum, in particular, is a model catalyst for both reactions because it catalyzes them efficiently, and is durable in the acidic environment of a PEM fuel cell, Abruña said.
But what about other materials?
Recent experiments with non-precious-metal HOR electrocatalysts aimed to overcome two major challenges, the researchers wrote: low intrinsic activity from too strong a hydrogen binding energy, and poor durability due to rapid passivation from metal oxide formation.
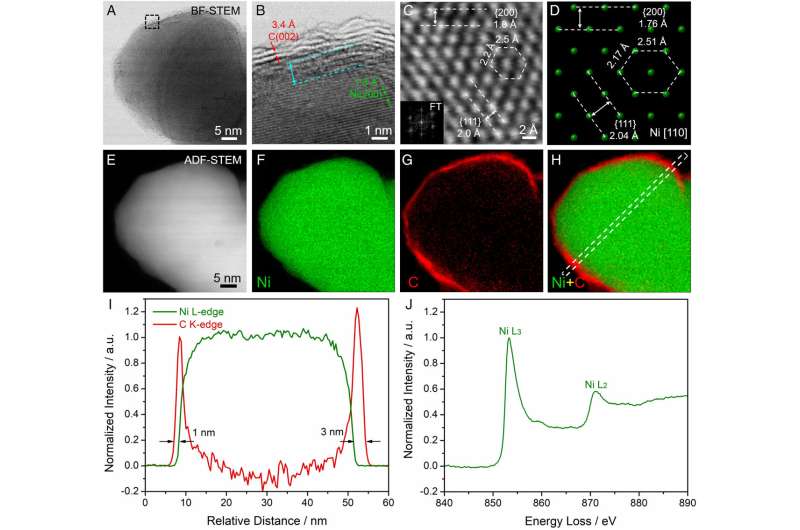
To overcome these challenges, the researchers designed a nickel-based electrocatalyst with a 2-nanometer shell made of nitrogen-doped carbon.
Their hydrogen fuel cell has an anode (where hydrogen is oxidized) catalyst consisting of a solid nickel core surrounded by the carbon shell. When paired with a cobalt-manganese cathode (where oxygen is reduced), the resulting completely precious metal-free hydrogen fuel cell outputs more than 200 milliwatts per square centimeter.
The presence of nickel oxide species on the surface of the nickel electrode slows the hydrogen oxidation reaction dramatically, Abruña said. The nitrogen-doped carbon coating serves as a protection layer and enhances the HOR kinetics, making the reaction quicker and much more efficient.
In addition, the presence of the graphene coating on the nickel electrode prevents the formation of nickel oxides—resulting in electrodes with dramatically enhanced lifetimes. These electrodes are also much more tolerant to carbon monoxide, which rapidly poisons platinum.
"The use of this novel anode would dramatically lower prices enabling the application of alkaline fuel cells in a wide variety of areas," Abruña said.
Co-authors include Francis DiSalvo, the John A. Newman emeritus professor of chemistry; Yao Yang, Ph.D. '21; David Muller, the Samuel B. Eckert Professor of Engineering in the College of Engineering and the co-director of the Kavli Institute at Cornell for Nanoscale Science, as well as collaborators from Wuhan University in the laboratory of Lin Zhuang and University of Wisconsin, Madison with Manos Mavrikakis.
In February, Abruña and colleagues, including DiSalvo, found that a cobalt nitride catalyst is nearly as efficient as platinum in catalyzing the oxygen reduction reaction.
More information: Yunfei Gao et al, A completely precious metal–free alkaline fuel cell with enhanced performance using a carbon-coated nickel anode, Proceedings of the National Academy of Sciences (2022). DOI: 10.1073/pnas.2119883119
Journal information: Proceedings of the National Academy of Sciences
Provided by Cornell University