Phase transformation in titanium alloys observed at atomic scale
Titanium is an important structural metal. Titanium alloys are widely used in aerospace and biomedical applications, thanks to their light weight, remarkable specific strength, superb corrosion resistance and high heat resistance.
Recently, Prof. Yu Qian in Prof. Zhang Ze' s group at the Center of Electron Microscopy in Zhejiang University led her team in collaboration with Prof. Ma En and Prof. Zhang Wei from Xi'an Jiaotong University and Prof. Long-qing Chen from the Pennsylvania State University to explore how titanium evolves in microstructure at a high temperature. By employing cutting-edge, in situ and multi-scale electron microscopy techniques as well as synchrotron radiation and computational simulations, the researchers together conducted an in-depth study into the α-β transformation mechanism in the titanium-molybdenum alloy. They discovered a significant distinction between this transformation process and what is described in classical nucleation theory. Their research findings appeared in the journal Nature Materials on November 26.
A phase in an alloy is usually a homogeneous component marked by the same aggregation state, crystal structure and property. Different phases have different features due to their difference in structure and composition. In materials' design, we can take full advantage of their complementary strengths so as to optimize the overall performance of the material.
Titanium-based alloys are usually the mixture of both the low-temperature hexagonal closed-packed (hcp) α-phase and the high-temperature body-centered cubic (bcc) β-phase. The combination of the dual phases is tuned by alloying of β-stabilizing (like Mo, Nb, Ta) and α-stabilizing (like O, Al, La) elements. The phase transformation can be plainly understood as the transition from one phase to another induced by external stimuli.
"To achieve the precise regulation of the phase structure, we need to know the real process of the phase transformation and its fundamental law," said Prof. Yu.
The classical nucleation and growth mechanism starts from the nucleation of a new phase, which assumes the same crystal structure and composition as the final equilibrium product phase. However, this simple picture fails to explain the phase transformation paths in many alloy systems. Often, the evolution into the product phase is preceded by one or more intermediate states that are distinctly different from the product phase, in terms of composition, crystal structure and chemical order. The intermediate state is metastable and, as it evolves, its thermodynamic properties change as well, including its interfacial energy with the surroundings. There have been recent reports suggesting non-classical nucleation during the formation of inorganic nanoparticles from solutions, crystallization of proteins16 and nucleation of diamond crystals as well as crystallization of amorphous tungsten carbides, but the atomic details of complex reactions in metallurgic systems remain elusive.
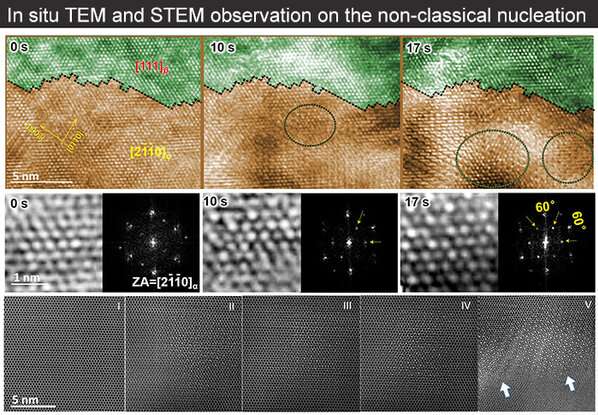
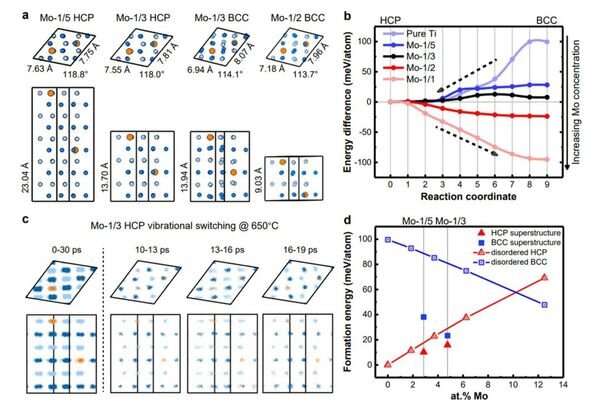
Yu Qian et al. conducted a direct and time-resolved experimental observation of a non-classical nucleation-mediated phase transformation at sub-ångström resolution using a titanium-molybdenum alloy as a representative system. They discovered a nano-sized and chemically ordered superstructure in the α-phase matrix; its composition, chemical order and crystal structure were all found to be different from both the parent and the product phases, but triggered an increasingly low energy barrier for the transformation into the β-phase. This latter phase transition can proceed instantly via vibrational switching when the molybdenum concentration in the superstructure exceeds a critical value.
"It is the first time that such a non-classical solid-state phase transformation has been reported in titanium-based alloys," said Prof. Yu. "Our study can be held up as a prime example where in-situ and atomic-scale experiments are combined with computer simulations. Therefore, it opens the door to research into solid-state phase transformations in other alloys."
More information: Xiaoqian Fu et al, Atomic-scale observation of non-classical nucleation-mediated phase transformation in a titanium alloy, Nature Materials (2021). DOI: 10.1038/s41563-021-01144-7
Journal information: Nature Materials
Provided by Zhe Jiang University