Lipid droplets as endogenous intracellular microlenses
With the demand in real-time monitoring of endoplasmic variations and rapid detection of extracellular signals, a great number of approaches to bioimaging have been developed. The past few decades have witnessed a dramatic progress in optical imaging, especially with the emerging of microsphere-assisted techniques that have the excellent ability of signal collection and enable real-time and super-resolution imaging with conventional optical microscopic systems. However, as most of the microspheres in current strategies are in solid and artificially synthetic materials, they are of very low biocompatibility. Fortunately, lessons from nature have shown that some bio-components and already-existing objects can interact with light and are able to take the same functions as the real optical elements.
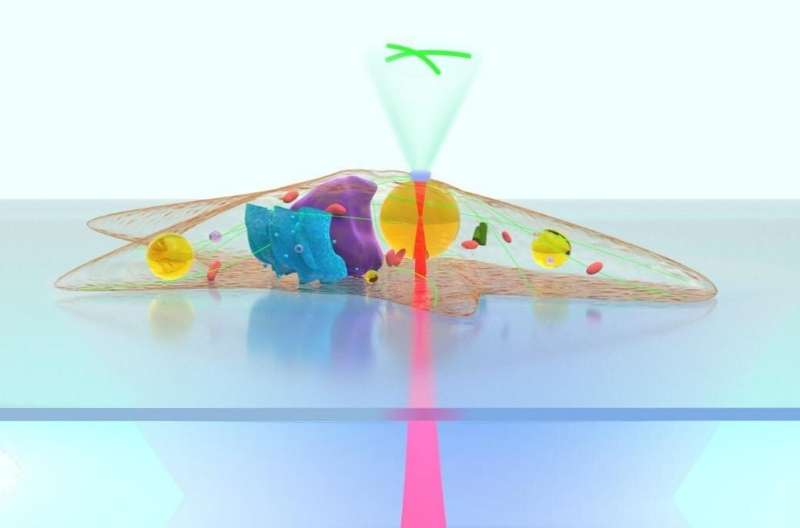
In a paper newly published in Light Science & Application, a team of researchers, led by Professors Baojun Li, Yao Zhang and Yuchao Li from Institute of Nanophotonics, Jinan University, China and Dr. Pietro Ferraro from CNR-ISASI, Institute of Applied Sciences and Intelligent Systems, Italy, demonstrate that lipid droplets, dynamic structures that naturally exist in cells, can act as intracellular microlenses for real-time monitoring of subcellular structures and detection of extracellular signals. With a spherical shape and a refractive index higher than cytoplasm and periplasm, the lipid droplets exhibit the lensing effect to efficiently converge both the excitation light and the fluorescence signals.
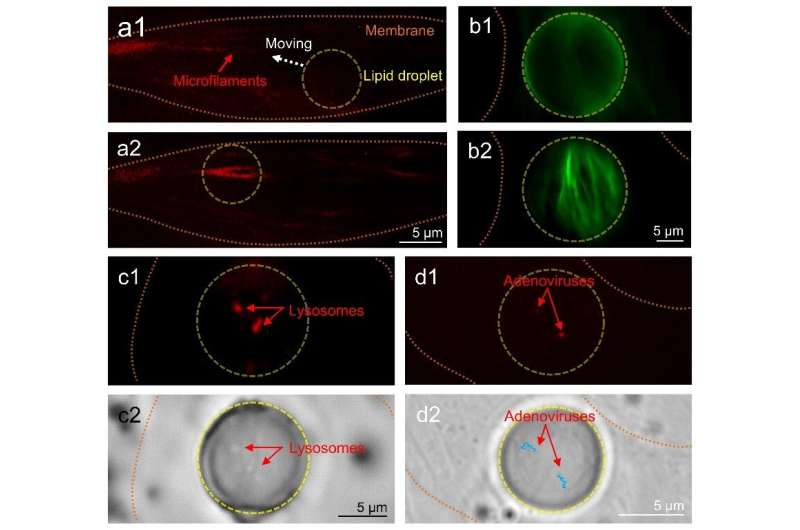
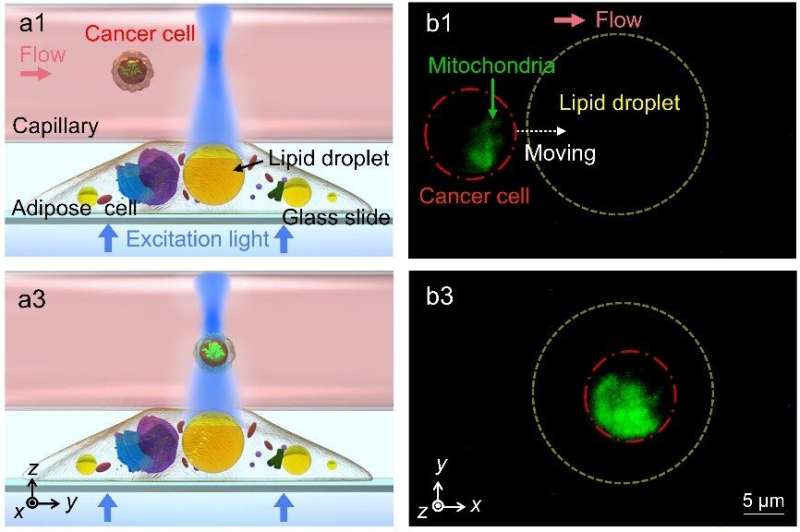
To achieve intracellular imaging of subcellular structures, the lipidic microlenses work in a contact mode in which the collection efficiency of the emitted fluorescent signals is greatly increased by the lipid droplets. As a result, the required excitation power in fluorescence imaging can be reduced by up to 73%. Driven by scanning optical tweezers, the lipid droplets can be moved inside the cell to perform an enhanced intracellular fluorescence imaging of the microfilaments, lysosomes and adenoviruses in living cells. Moreover, due to the refractive index contrast of the lipid droplets to cytoplasm, the long focal length of the lipid droplets can extend the fluorescence enhancement strategies to a non-contact mode that increases the working distance of imaging and detection by the lipidic microlenses. In the non-contact mode, the excitation light is highly converged by the lipid droplets to enhance the fluorescence intensity from the extracellular environment surrounding the cells. In the experiments, an efficient detecting of fluorescent signals of cancer cells in extracellular fluid was accomplished by the lipidic microlenses.
The presented lensing effect of the lipid droplets is expected to find applications with fully biocompatible miniaturized tools for biosensing, endoscopic analysis and single-cell diagnosis. The use of the lipid droplets as intracellular microlenses also provides opportunities to design and construct diverse endogenous photonic devices.
More information: Xixi Chen et al, Lipid droplets as endogenous intracellular microlenses, Light: Science & Applications (2021). DOI: 10.1038/s41377-021-00687-3
Journal information: Light: Science & Applications
Provided by Chinese Academy of Sciences





















