New details behind how the Shigella pathogen delivers bacterial proteins into our cells
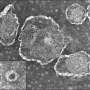
Shigella, a bacterial pathogen that causes dysentery and is the leading cause of childhood diarrheal diseases, inserts a pore called a translocon into an infected person's intestinal cells and then injects bacterial proteins into the cells. There, the proteins hijack the cells' machinery to help Shigella multiply. In a study published in mBio, a team at Massachusetts General Hospital (MGH) has uncovered important details about Shigella's translocon, which may help researchers develop an effective strategy to block this critical component of infection.
"Shigella infects our gut by manipulating our intestinal cells and tricking them into letting the Shigella inside. In fact, there are many bacterial pathogens that use this same, or similar, mechanism to infect us," says lead author Poyin Chen, Ph.D., a postdoctoral fellow at MGH. "This translocon pore is essentially the gateway through which bacterial proteins get pumped into our cells. We know that this structure is made of two proteins—IpaB and IpaC—but what we don't know is how these proteins fit together to make this pore."
When the investigators used protein mapping techniques to look closely at translocons when they were embedded in cell membranes, they were able to see which of the two proteins—specifically IpaB—makes up the inner ring of the pore. "If you think of the translocon pore as a donut, this would be the walls of the donut hole. This finding is important because this is the part of the translocon pore that directly interacts with bacterial proteins as they are injected into our cells," explains Chen. "With the findings from this study, we can begin to understand if this pore acts as a slippery tube that bacterial proteins travel through or if the translocon pore can control the flow of bacterial proteins into our cells."
Such details may help investigators target the translocon and block the entry of Shigella proteins into cells. "For something that is so essential to establishing infection, we know terribly little of how it's made and how it works," says Chen. "As we gain a better understanding of its parts, we will be able to approach the structure as a whole and maybe even find ways to neutralize the function of this structure to prevent infection before it can begin."
Co-authors include Brian C. Russo, Jeffrey K. Duncan-Lowey, Natasha Bitar, Keith Egger and Marcia B. Goldberg.
More information: Topology and contribution to the pore channel lining of plasma membrane embedded S. flexneri type 3 secretion translocase IpaB, mBio (2021).
Journal information: mBio
Provided by Massachusetts General Hospital