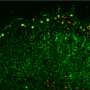
Septin proteins act as cellular police to identify, imprison and kill 'superbug' Shigella
Using state-of-the art technologies to image human cells and study infection at the level of a single bacterial cell, the research team, led by the London School of Hygiene & Tropical Medicine, has uncovered the strongest evidence yet that septins take Shigella prisoner.
Crucially, it reveals for the first time that these proteins can detect where bacteria will split for division and prevent it from doing so by forming cage-like structures around the bacteria.
Antimicrobial resistance is one of the biggest threats to global health. As well as the need to develop new drugs, such as antibiotics, novel ways to control bacterial infection are vitally important. Shigella is a human gut pathogen, infects more than 150 million people globally and causes up to 500,000 deaths every year. Due to the increasing number of drug resistant strains, Shigella is one of the 'superbugs' deemed a priority by the World Health Organization.
The research team say that although septins are a powerful, natural mechanism to restrict Shigella, future work is required to determine how septin biology can be harnessed for therapeutic purposes. It is hoped that these new findings may lead to a novel way to boost the human immune system and treat a wide variety of bacterial infections.
Lead author Professor Serge Mostowy from the London School of Hygiene & Tropical Medicine said: "We are actively working to engineer this discovery for human health application. If we can use drugs to boost septin caging, we have a new way to stop infection."
In 2010, researchers first observed that septin cages can entrap Shigella, opening up the tantalising prospect of a new way to stop the bacteria spreading in the body. However, how cells recognise Shigella for entrapment, and the fate of entrapped bacteria, was mostly unknown.
Study co-author Sina Krokowski said: "For modern medicine, how cells can recognise bacteria is the subject of intense investigation. This information is crucial if septins are ever able to be used as a treatment for humans."
Excitingly, the research team found that the septin cage seems to recognise actively dividing bacteria, whether it's antimicrobial resistant or not. Moreover, using high resolution microscopes with state of the art cameras, the research team found that once entrapped in a septin 'cage', 93% of bacteria will never divide again because they are targeted to autophagy, a cellular process of 'self eating', providing definitive proof that cages are anti-bacterial.
Professor Mostowy said: "The rise of 'superbugs' is one of the greatest global health challenges we face. New drugs to tackle antimicrobial resistance are crucial but they are costly and all likely to be met with resistance. We must therefore also look at other, novel ways to control bacterial infection.
"By applying cutting edge microscopy techniques, only available in the last few years, to study the cellular immune response to Shigella, we now have clear evidence that septins can be a new 'natural' weapon in the fight against AMR. Remarkably, these proteins act as host cell 'sensors' to recognise actively dividing bacteria, the exact bacterial population that causes disease, for entrapment. In addition to Shigella, this may also apply to a wide variety of invasive bacterial pathogens such as Pseudomonas and Staphylococcus."
The authors acknowledge limitations of the study including the possibility that some bacteria have evolved to avoid septin cage entrapment, and the need for in vivo study prior to application in humans.
More information: Sina Krokowski et al, Septins Recognize and Entrap Dividing Bacterial Cells for Delivery to Lysosomes, Cell Host & Microbe (2018). DOI: 10.1016/j.chom.2018.11.005
Journal information: Cell Host & Microbe
Provided by London School of Hygiene & Tropical Medicine