New carbonyl rearrangement method could help identify new medicines, chemicals

Rearranging the parts of a molecule is like rearranging the contents of a tightly packed suitcase: You must go through a number of steps, taking objects in and out to get the configuration you want.
A new study by University of Chicago chemists, however, offers a more efficient way to let researchers rearrange the components of a molecule—in particular, a key component called carbonyl groups that appears in many pharmaceutical drugs and other useful chemicals.
"This method really simplifies the modification of an important functional group in organic chemistry," said Zhao Wu, a postdoctoral researcher and first author of the paper, published Nov. 5 in the journal Science.
Carbonyl groups consist of a carbon atom double-bonded to an oxygen atom. These components appear in many useful molecules, including pharmaceutical drugs, and researchers would like to test them in many different configurations to find which is best. "The location of the group on the molecule is really important for biological activity," said Prof. Guangbin Dong, the study's corresponding author.
A carbonyl group located along the outside of a molecule, for example, may help the molecule latch onto the correct cells in the liver for a drug to treat diabetes. But a researcher who wants to try that design can't just unplug the carbonyl group and plug it into another spot: in the end, it often takes five or more steps to move it even just a few atoms over. Having to repeat this experimental process over and over for each new compound slows down the pace of discovery.
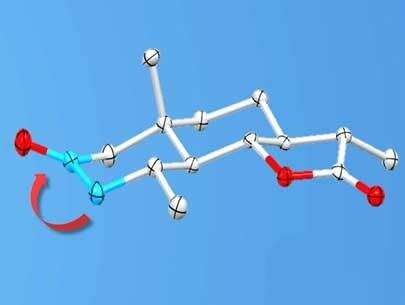
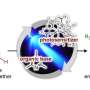
Building on their previous work, the UChicago team was instead able to design a method that uses two separate catalysts, which work together to allow selective chemical reactions. This sidestepped a common problem with such reactions, where side reactions start occurring alongside the desired ones; those side reactions can generate unwanted byproducts and shut down the catalyst's ability to continue boosting the primary reactions.
In all, the team was able to reduce the number of steps to only one or two. They also found that the new method improved selectivity—the ability to choose precisely where the carbonyl group ends up.
"This method should make it much easier to produce new molecules for drug discovery, and perhaps also synthesizing other natural products," said Wu.
The scientists plan to continue working to improve the process, to streamline steps, boost efficiency and expand the number of different substrates that can be used. "This is just the beginning; there are many ways to further develop this method," said Dong.
More information: Zhao Wu et al, Carbonyl 1,2-transposition through triflate-mediated α-amination, Science (2021). DOI: 10.1126/science.abl7854
Journal information: Science
Provided by University of Chicago