Enabling selective carbon-carbon bond formation at the inherently inert position of enol silyl ethers
Nagoya University researchers have discovered a new way to modify reactants that have been widely studied and utilized for over 50 years, enabling more efficient production of some complex organic molecules used in medical drugs. The work was published in Nature Communications.
Synthetic chemists often use a class of molecules called enol silyl ethers, in which silicon atom (Si), oxygen atom (O), and carbon atom (C) are connected as Si-O-C=C (- is a single bond, and = is a double bond). These molecules are reactive to form a new bond at the carbon atom not attached to the oxygen atom, serving as reliable substrates for efficient and selective synthesis of functionalized carbonyl compounds, and thus has been widely utilized in synthetic organic chemistry over 50 years.
Professor Takashi Ooi and colleagues from Nagoya University's Institute of Transformative Bio-Molecules in Japan wanted to find out if selective C-C bond formation at an inherently inert position of enol silyl ethers could be accomplished to form more complex enol silyl ethers, which are then available for further chemical transformations.
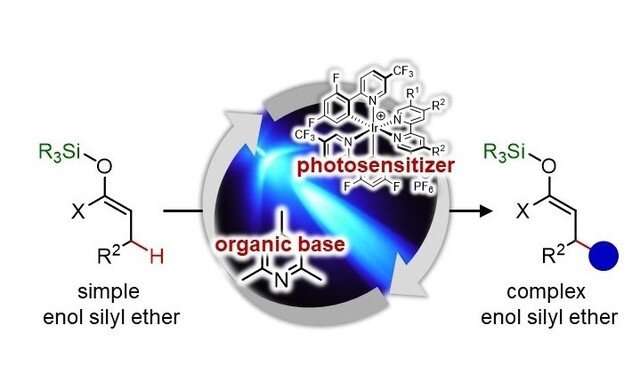
They developed a chemical reaction by the combined use of two catalysts, a photosensitizer and an organic base, under visible-light irradiation (blue LED). The new reaction does not eliminate a silyl group, which normally leads to the formation of simple carbonyl compounds. Rather, it cleaves a relatively stable carbon-hydrogen bond, which enables the replacement of the hydrogen with an alkyl group. The new process allows chemists to synthesize previously difficult-to-access complex carbonyl compounds, and it can accelerate the discovery of new drugs.
This catalytic strategy is expected to help streamline synthesis of a variety of other organic molecules as well. "Our strategy is not limited to enol silyl ethers," says Ooi. "We would like to apply it to other organic compounds to enable bond-forming reactions at previously difficult-to-functionalize positions in molecules."
The article, "Direct allylic C-H alkylation of enol silyl ethers enabled by photoredox-Brønsted base hybrid catalysis," was published in Nature Communications.
More information: Kohsuke Ohmatsu et al. Direct allylic C–H alkylation of enol silyl ethers enabled by photoredox–Brønsted base hybrid catalysis, Nature Communications (2019). DOI: 10.1038/s41467-019-10641-y
Journal information: Nature Communications
Provided by Nagoya University