Chemists propose new beneficial catalyst for initial materials in pharmacy

A collaboration of researchers from RUDN University (Russia), Centro de Química Estrutura (Portugal) and Baku State University (Azerbaijan) proposes a new potential method to produce initial compounds for many chemical industries, including pharmacy, cosmetics, dyes and liquid crystal production. The new method of room-temperature synthesis has high yields, and is described in two articles published in the Journal of Organometallic Chemistry and in Inorganica Chimica Acta.
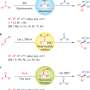
Cyanosilylation is the addition of cyanogroups—NΞC to silyl groups, which include silicon and another inconstant radical component, labeled with 'R' in the scheme below. Asymmetric addition of these groups to chemical compounds with =C=O groups leads to the formation of a variety of enantiomerically enriched compounds. These are new prospective materials and initial substances for the chemical industry.
Enantiomers are optical isomers that contain the same atoms and that are mirror images of each other, but are nonetheless not identical and may have different chemical and physical properties. It is these properties that make them so promising. The process of cyanosilylation is driven by additional agents called catalysts. Catalysts mediate the reaction path, but they do not change themselves, and quit the process without any transformations at the end. Catalysis allows the reaction to proceed faster and more intensively, even at lower temperatures or over a wider pressure range.
Catalytic asymmetric cyanosilylation has been extensively studied in recent years due to the significant utility of the products of this reaction—cyanohydrin trimethylsilyl ethers. They are the key starting materials in the syntheses of substantial chemical products, e.g., α-hydroxy aldehydes, α-hydroxy acids, β-amino alcohols, etc. The first group is used in colourants, pharmaceutical, fungicide and odourant synthesis, the second is essential for food production and pharmaceuticals, and the third group is useful for the household chemicals and cosmetics. It's notable that all these compounds are easy to produce from this type of ethers through only one-step synthesis.

The main synthetic route for the preparation of cyanohydrin trimethylsilyl ethers is the addition reaction of trimethylsilyl cyanide (TMSCN) that contains both the cyanogroup and the silyl component to carbonyl compounds (those with =C=O groups, as mentioned earlier). Trimethylsilyl cyanide is a source of cyanide that is safe and easy to handle, if compared to HCN, NaCN and KCN.
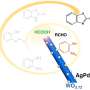
"Currently, a variety of organo- and metal catalysts have been employed in this reaction. These protocols have many disadvantages, such as low yield, long reaction time, harmful solvents, etc. Thus, in this project we have introduced new types of catalysts for cyanosilylation of aldehydes," says Atash Gurbanov, the leading author of the paper. The candidate is arylhydrazone in complex with Cu(II) and Co(II/III). Apart from the radicals, arilhydrazone contains an aril group (an aromatic ring lacking one hydrogen) and two atoms of nitrogen, one of which is in double chemical bond with carbon. Arilhydrazones are easy to produce and they are extensively studied by chemists.
These complexes act as homogeneous catalysts for the cyanosilylation of aldehydes (both aromatic ones and those without aromatic rings) with trimethylsilyl cyanide. The researchers argue that this reaction proceeds even "at room temperature in methanol, providing cyanohydrin trimethylsilyl ethers with good yields." The researchers say they have developed methods to promote the reaction yield for those two groups of aldehydes.
More information: Atash V. Gurbanov et al, Trinuclear and polymeric cobalt(II or II/III) complexes with an arylhydrazone of acetoacetanilide and their application in cyanosilylation of aldehydes, Inorganica Chimica Acta (2017). DOI: 10.1016/j.ica.2017.07.004
Journal information: Journal of Organometallic Chemistry , Inorganica Chimica Acta
Provided by Peoples' Friendship University of Russia