The next big thing: How do scientists bring hydrogen fuel cells from the laboratory to public life?
Fuel cells, due to their high efficiency and environment-friendly attributes in the process of electricity generation, are gaining popularity for Fuel Cell Vehicle (FCV) production, such as automobiles, forklifts, buses and airplanes. However, the costly nature of producing fuel cell catalysts precludes the mass-production and large-scale application of FCVs.
Fuel cell catalysts are usually made of platinum (Pt) or Pt alloys with transition metals thinly coated onto the porous carbon supports. Platinum is an ideal catalytic material as it can withstand the acidic conditions and increase the rate of chemical reactions efficiently. However, it is expensive and has insufficient resource reserves. Therefore, it is imperative to develop and screen new catalysts with low Pt quantity and high catalytic activity for fuel cell commercialization.
In a Science paper published on Oct. 22, researchers at the University of Science and Technology of China (USTC) of the Chinese Academy of Sciences (CAS) reported a sulfur-anchoring method of high-temperature, successfully synthesized small-sized Pt intermetallic nanoparticle (i-NP) catalysts with ultralow Pt loading and high mass activity. They also established i-NP libraries, including 46 types of Pt nanoparticles (NPs) to screen inexpensive and durable electrode materials as well as explore structure-activity relations of i-NPs systematically.
I-NPs have attracted wide attention because of their unique atomically ordered properties and excellent catalytic performance in many chemical reactions. However, inevitable metal sintering at high temperature is undesired during the synthesis of i-NPs, as it will lead to larger crystallites. Thus, it results in a decreased specific surface area and lower catalytic activities of the materials, and eventually reduces the utilization rate of Pt, therefore greatly increasing the cost of fuel cells.

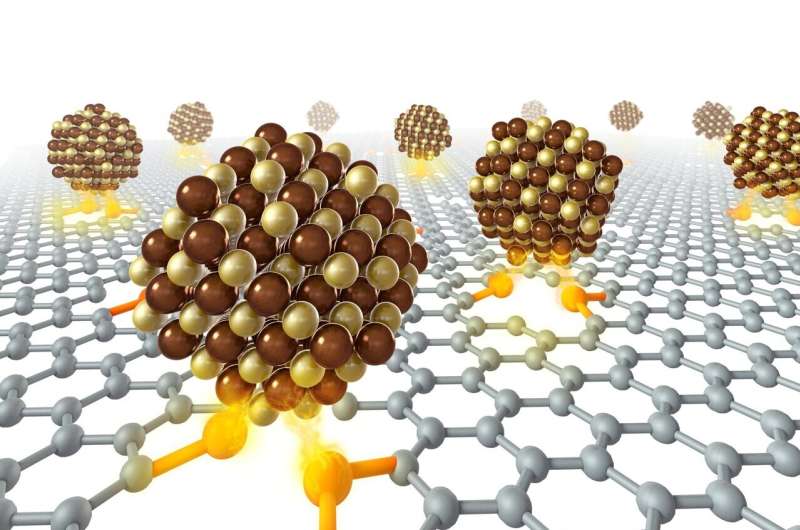
The research team, led by Liang Haiwei, ingeniously utilized strong Pt-sulfur chemical interaction. They prepared Pt intermetallics on sulfur-doped carbon (S-C) supports in order to suppress NPs sintering at high temperatures, and they were able to obtain atomically ordered i-NPs with an average size of <5 nm. S-C supports showed excellent anti-sintering ability, and researchers obtained Pt NPs with the average diameter still <5 nm after annealing at high temperatures up to 1000 C. However, severe Pt sintering was observed after the same annealing process on commercial carbon black supports.
To take advantage of the anti-sintering property, researchers synthesized 46 types of small-sized Pt-based i-NPs on S-C supports and established i-NP libraries. Spectral characterizations were measured, and the results verified the strong chemical interactions of Pt-S bonds. Moreover, the X-ray diffraction (XRD) results showed a high ordering degree and small size of i-NP catalysts in libraries, consistent with the statistical analysis of the high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) observations.
-

Schematic illustration of the high-temperature sulfur-anchoring synthetic approach. Credit: LIU Xinyi, LIU Zige -

Schematic illustration of the high-temperature sulfur-anchoring synthetic approach. Credit: LIU Xinyi, LIU Zige
"Based on the i-NP libraries, we can systematically study the relationship between structure and performance of catalysts," said Liang, "and sufficient samples helped us screen out efficient catalysts which were expected to largely decrease the cost of fuel cells." Researchers screened i-NPs and applied them for proton-exchange membrane fuel cells (PEMFCs). These catalysts exhibited excellent electrocatalytic performance for oxygen reduction reaction (ORR). Especially in H2-air PEMFC, although the Pt loading of i-NPs was 11.5 times lower than that of Pt/C cathode, the i-NP catalysts' cathodes showed similar ability to the Pt/C cathode.

This work provides a universal way for the synthesis of Pt alloy catalysts utilized in hydrogen fuel cells. This method raises hopes for reducing the quantity of Pt used, thereby decreasing the cost of fuel cells. "By engineering the porous structures and surface functionalities of carbon supports, the efficiency of fuel cells can be further improved, thus accelerating their transferrance from laboratory to the public," said Liang.
More information: Nikita Hanikel et al, Evolution of water structures in metal-organic frameworks for improved atmospheric water harvesting, Science (2021). DOI: 10.1126/science.abj0890. www.science.org/doi/10.1126/science.abj0890
Journal information: Science
Provided by University of Science and Technology of China