Controlling the length of supramolecular polymers
Systems made up of one supramolecular polymer are well understood, but much remains unknown for systems involving the combination of multiple supramolecular polymers, such as what affects the length of the resulting copolymers. For her Ph.D. research, Elisabeth Weyandt examined how the length of these copolymers change. Weyandt defended her thesis at the department of Chemical Engineering & Chemistry on September 17.
In conventional polymers, like those used in plastics, monomers are joined together by covalent bonds. However, in supramolecular polymers things are a little different as monomers are connected by a combination of weaker, non-covalent bonds like hydrogen bonding or π-π stacking.
Single component supramolecular polymer systems are well understood but moving from single to multi-component systems to obtain functional supramolecular materials and systems remains challenging. When mixing several supramolecular components, the aggregate length is affected, which in turn affects material properties.
Adding a second supramolecular polymer
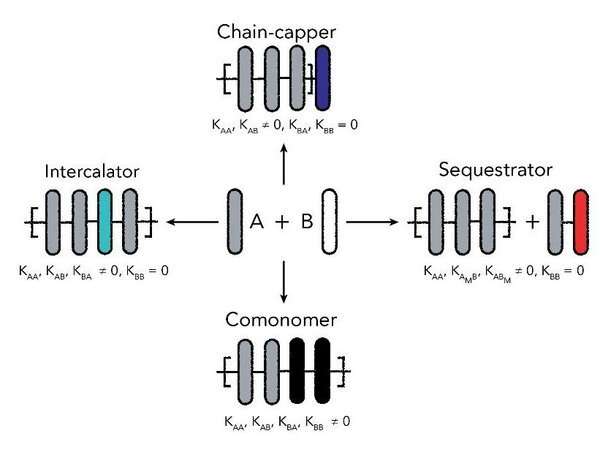
For her Ph.D. research, Weyandt explored for the first time how adding a second supramolecular polymer affects the resulting copolymer length. The second component can have a number of effects. First, it could act as a comonomer that increases the length. Second, it can act as an intercalator that squeezes between two monomers. Third, it can behave as a sequestrator that negatively impacts single monomers and decreases the number of available monomers. Finally, it can act as a chain-capper that blocks the chain end and stops any increase in length of the polymer.
In her thesis, Weyandt outlines the difficulties encountered in measuring the length of the dynamic polymers, as well as analyzing the role that the additive plays in the process. A key finding from the research was that the total monomer concentration has a significant influence on how the second polymer controls the length of the copolymer. When operating at high concentrations, the efficiency is higher than at low concentrations, which is counterintuitive given that the composition of monomers is exactly the same.
Light irradiation
Weyandt studied several cases with supramolecular mixtures. In one case, she uses a photoswitchable additive, which is an additive that can change from an intercalator to a chain-capper by irradiating it with light. The additive was then changed back to an intercalator by thermal relaxation.
The non-covalent bonds formed between supramolecular monomers give rise to their unique, dynamic properties; but also bring many challenges in resolving the mechanism behind these changes. With the work in her thesis, Weyandt has broadened the fundamental understanding of supramolecular copolymers and the subtle balance of interactions that drive the structure formation in complex, multi-component supramolecular systems.
More information: Gijs M. ter Huurne et al, Controlling the Length of Cooperative Supramolecular Polymers with Chain Cappers, Chemistry – A European Journal (2020). DOI: 10.1002/chem.202001293
Journal information: Chemistry – A European Journal
Provided by Eindhoven University of Technology