An efficient method for separating O-18 from O-16, essential for use in cancer treatment
Positron Emission Tomography (PET) plays a major role in the early detection of various types of cancer. A research group led by Specially Appointed Professor Katsumi Kaneko of the Research Initiative for Supra-Materials (RISM), Shinshu University have discovered a method to separate oxygen-18 from oxygen-16, an essential isotope for PET diagnosis, at high speed and high efficiency. The results of this research were recently published online in the journal Nature Communications.
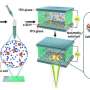
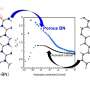
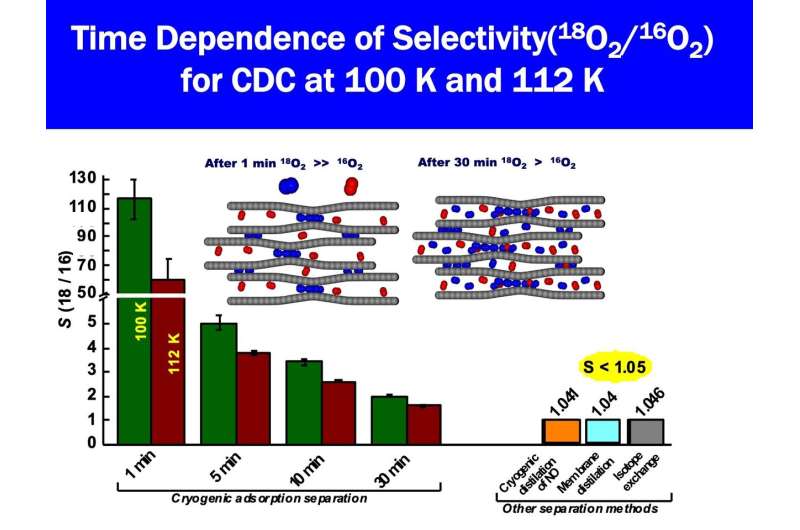
The novel method for the rapid and efficient separation of 18O from 16O2, which is abundant in the atmosphere, was carried out with nanoporous carbon, which is made of pores smaller than 1 nanometer. When a mixture of 16O2 and 18O2 is introduced into the nanoporous carbon, the 18O2 is preferentially adsorbed and is efficiently separated from 16O2. The experimental separation of 18O2 from 16O2 was also conducted using the low-temperature waste heat from a natural gas storage facility.
18O plays a major role in the early detection of cancer. Taking advantage of the property of cancer cells which take up much more glucose than normal cells, doctors inject a drug called 18F-FDG (fluorodeoxyglucose), which is an index of glucose metabolism and uses a PET machine to clarify which part of the body has cancer. 18F-FDG is a drug in which fluorine-18 (18F), which emits positive electricity, is attached to glucose. 18F-FDG is produced by a nuclear reaction in which 18O is introduced before the protons are injected. Therefore, 18O is an important substance indispensable for PET diagnosis but was difficult to procure because only 0.2% of naturally occurring oxygen is O-18. In order to separate 18O from the majority of 16O found in the atmosphere, it was necessary to distill 18O from 16O, even though they have very similar boiling points. This distillation required precise technology and took more than 6 months to complete.
The novel method using nanoporous carbon to distill 18O can be used not only for PET diagnosis but for research on dementia, and this novel method can be applied to the separation of carbon and nitrogen isotopes, and other molecules useful for isotopic analysis methods and therapeutic cancer drugs. The group expects more demand for this method and substance in the future.
More information: Sanjeev Kumar Ujjain et al, Adsorption separation of heavier isotope gases in subnanometer carbon pores, Nature Communications (2021). DOI: 10.1038/s41467-020-20744-6
Journal information: Nature Communications
Provided by Shinshu University