Researchers tailor rapid and effective N-phosphopeptides enrichment strategy
Protein N-phosphorylation plays a critical role in central metabolism and two/multicomponent signaling of prokaryotes. Furthermore, it exists as intermediates of some important enzymes in mammals.
However, the current enrichment methods for O-phosphopeptides are not preferred for N-phosphopeptides due to the intrinsic lability of P-N bond under acidic conditions. Therefore, the effective N-phosphoproteome analysis remains challenging.
Recently, a research group led by Prof. Zhang Lihua and Pro. Zhang Yukui from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) proposed a rapid and effective N-phosphopeptides enrichment strategy.
Their study was published in Nature Communications on Dec. 4.
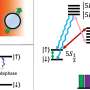
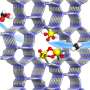
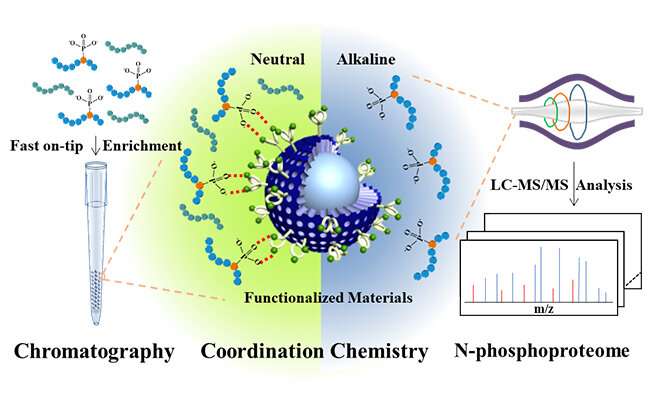
Inspired by naturally occurring phosphatases, which provide specific Zn(II)-central enzymatic pockets to bind phosphate units of substrates, the researchers designed bis(zinc(II)-dipicolylamine) molecular (DpaZn) for phosphate targets recognition under neutral conditions.
Combing the fast mass transfer ability of sub-2 μm core-shell silica microspheres, DpaZn-functionalized sub-2 μm core-shell silica microspheres (SiO2@DpaZn) were designed for on-tip N-phosphopeptides enrichment under neutral conditions. In total, 3,384 N-pho sites, containing 611 pHis, 1618 pLys and 1155 pArg, were identified from HeLa cell lysates.
The new N-phosphorylated proteome analysis method provides not only basic data for in-depth study of its biological function, but also technical support for promoting the development of precision medicine, synthetic biology and other fields.
More information: Yechen Hu et al. Bis(zinc(II)-dipicolylamine)-functionalized sub-2 μm core-shell microspheres for the analysis of N-phosphoproteome, Nature Communications (2020). DOI: 10.1038/s41467-020-20026-1
Journal information: Nature Communications
Provided by Chinese Academy of Sciences