Scientists identify electronic and structural dynamics of catalytic centers in single-Fe-atom materials
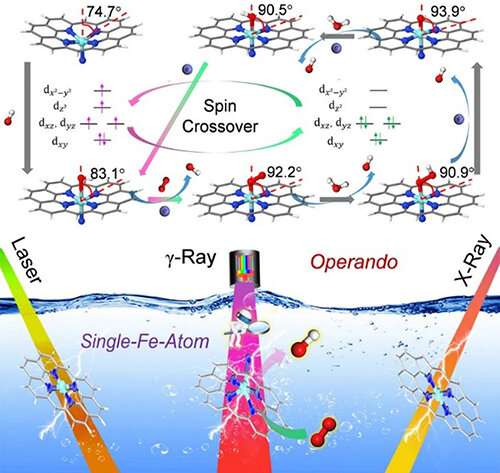
Single-atom catalyst (SAC) is a conceptual bridge between homo- and heterogeneous catalysis. It offers new opportunities for capturing the reaction intermediates by identifying the active sites, and even monitoring the dynamic behaviors of both the geometric structure and electronic environment of the catalytic sites at atomic scale.
Both the exploration of model SACs with a definite microenvironment and the development of novel operando techniques with high atomic resolution are significant, yet still challenging.
Recently, a research team led by Prof. Huang Yanqiang and Prof. Zhang Tao from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences and their collaborators reported their findings on electronic and structural dynamics of catalytic centers in single-Fe-atom materials under in situ oxygen reduction reaction (ORR) conditions.
The study was published in Chem on Nov. 20.
The researchers synthesized single-Fe-atom catalysts with well-controlled site density and definite N-coordination environment via a solvent-assisted linker exchange (SALE) method. They used it as the model system for electronic-level understanding of the ORR mechanism.
"We developed Operando Mossbauer spectroscopy for the first time to identify the exact structures and spin state of active atomically dispersed Fe moieties during ORR," said Prof. Huang.
By combining with operando Raman and X-ray absorption spectroscopy (XAS) measurements, as well as density functional theory (DFT) studies, the researchers proved that the potential-relevant structural and electronic evolutions of the active single-Fe-atom moieties were via directly capturing the *O2- and *OH-intermediates under ORR conditions.
These results provided a proof of concept for the integration of operando techniques and SACs, which might direct the way toward the electronic-level insight into the catalytic centers and reaction mechanism.
More information: Xuning Li et al. Identification of the Electronic and Structural Dynamics of Catalytic Centers in Single-Fe-Atom Material, Chem (2020). DOI: 10.1016/j.chempr.2020.10.027
Journal information: Chem
Provided by Chinese Academy of Sciences