Scientists capture the moments of nascent crystal formation and growth

Conversion of most materials into organized crystalline structure starts with the nucleation process. One everyday example is the rapid crystalization of supercooled water after the nucleation of a seed crystal. This phenomenon has perplexed scientists and ordinary people alike. The nucleation process, in which the atoms gather and form the smallest crystals, has been an important scientific phenomenon that has been widely studied since the late 1800s.
The classical nucleation theory states that the assembly of monomers into a crystal structure occurs in a one-directional fashion. On the other hand, there have been some who suggested that a non-classical crystallization process involving metastable intermediate crystal structures may occur in some systems. However, it has been extremely difficult to confirm these theories through direct observation because the nucleation occurs very rapidly, and the size of a nucleus can be as small as a few atoms.
This century-old mystery has been finally solved by an international joint research team led by LEE Won Chul, Professor of Mechanical Engineering at Hanyang University Erica Campus. The joint research team has succeeded in observing the moment of the initial state of nanocrystal nucleation.
The scientists succeeded in filming the process where the gold atoms gather to form nanocrystals. To observe the initial state of the nucleation process, the team synthesized gold nanocrystals by emitting electron beam onto gold cyanide nanoribbons on top of a graphene membrane, which decomposes the nanoribbons into gold atoms. The synthesized specimen was observed with the high-performance transmission electron microscope (TEM) at the Lawrence Berkeley National Laboratory. The process was recorded at an atomic level spatial resolution and an ultra-high temporal resolution on a scale of milliseconds.
The TEM observation showed the abrupt disappearance and reappearance of crystal lattice structures prior to the emergence of a stable crystal structure. Through careful analysis, the team ruled out some factors which may result in such observations such as the orientation, tilt, and fast rotation of nanocrystals. Therefore, the observed results appeared to indicate that the atoms making up the nucleus randomly oscillate between the disordered and crystalline states. This structural fluctuation appeared to occur spontaneously in a stochastic manner. The team's discovery directly challenged the longstanding nucleation theory as well as a more recent nucleation theory that has been proposed in the last two decades.
In addition, the team found that the stability of the crystalline state increased as the size of the nanocrystals increased. For example, the nanocrystals with 2.0 nm2 areas spent approximately half of the time existing in a crystalline state. When the crystal sizes increased to above 4.0 nm2 in area, the crystals appeared to exist most of the time under a crystalline form.
-
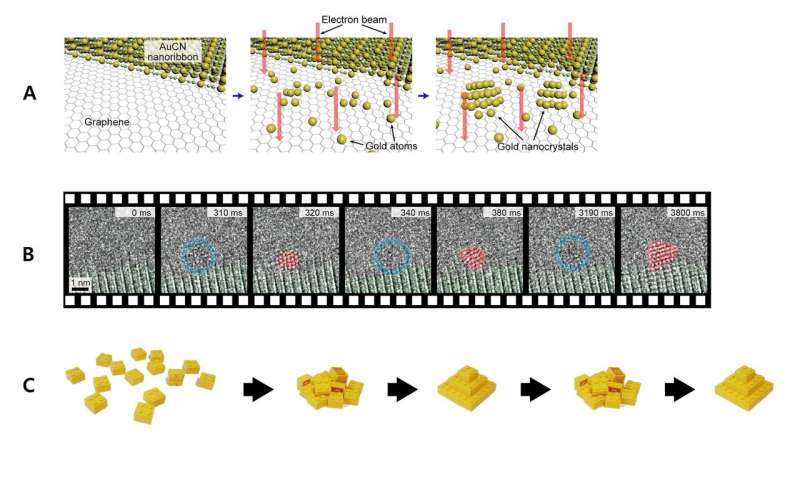
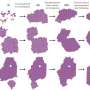
A) The schematics of the experiment. The AuCN nanoribbons on top of a graphene membrane were irradiated with electron beams. This decomposes the ribbons to generate gold atoms, which subsequently nucleate into nanocrystals. B) Still frames of the TEM video at various points of the nucleation process. C) Lego block model representation of transition of the gold nanocrystal structure between disordered and crystalline states. Credit: Institute for Basic Science -
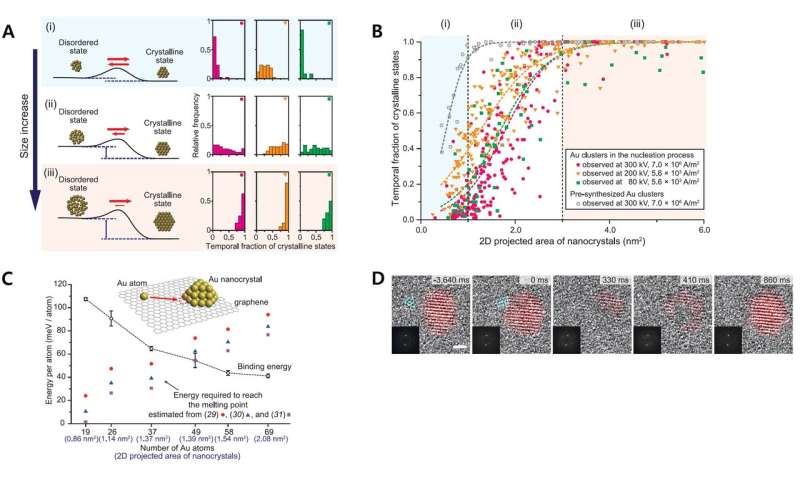
A) The new thermodynamic theory behind the nucleation process that the team proposed. The energy barrier between disordered and crystalline state is relatively low when the structure has relatively fewer atoms. The energy barrier increases and the crystalline state becomes more stable as the crystal size increases. B) The fraction of time in which the atoms exist under crystalline state versus the area of the nanocrystals. C) The energy required to reach a depressed melting point versus the number of gold atoms within the crystal. D) Merger between smaller and larger nanocrystals temporarily converts the entire structure back to a disordered state. Credit: Institute for Basic Science
In order to describe this phenomenon, the team proposed a new thermodynamic theory of crystal nucleation. The study proposed that the energy barrier between crystalline to disordered transformation tends to be very low in the earliest stage of nucleation when the cluster size is small and that it increases as more atoms are added to the structure. This can explain the spontaneous fluctuation between crystalline and disordered states in nascent crystals consisting of a few atoms. The team also pointed out in relatively smaller nanocrystals, even the addition of extra atoms can transfer enough energy into the system to transform the entire structure back to a disordered state. The energy barrier increases as the crystal grows, which reduces the probability of spontaneous reversion and stabilizes the crystalline structures in larger crystals.
Regarding these findings, Prof. Jungwon Park said, "From a scientific point of view, we discovered a new principle of crystal nucleation process, and we proved it experimentally."
Prof. Won Chul Lee said, "In an engineering point of view, by reproducing the initial state of the deposition process, it can be used to achieve original technology in semiconductor materials, components, and equipment."
This research was published in the journal Science on January 29, 2021.
More information: "Reversible disorder-order transitions in atomic crystal nucleation" Science (2021). science.sciencemag.org/cgi/doi … 1126/science.aaz7555
Provided by Institute for Basic Science