High pressure causes hydrogen variants to collapse
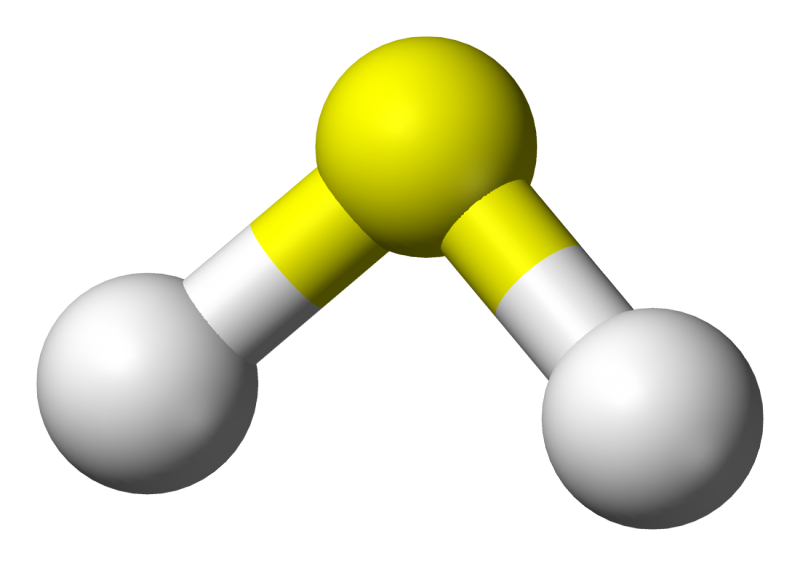
Hydrogen exists as a gaseous compound of two hydrogen atoms (H2). Under normal laboratory conditions, H2 occurs in the variants "ortho hydrogen" and "para hydrogen." Until now, it has been unclear how these variants behave under very high pressure. Researchers at the University of Bayreuth have now found the answer. Both ortho- and para-hydrogen become unstable under high pressure and cease to exist as distinguishable states. The research results presented in Nature Communications extend our physical understanding of fundamental quantum mechanical processes.
The two states of molecular hydrogen, ortho and para hydrogen, are known in research as spin isomers. They have the same chemical structure, but differ in the way the nuclei of the "twin atoms" connected in an H2 molecule relate to each other in terms of their angular momentum. This results in different physical properties of the spin isomers, for example differences in electrical and thermal conductivity. The question of whether spin isomers coexist under very high pressures is of great interest for planetary research and also for the fundamentals of quantum mechanics. Gas giants such as Jupiter contain large amounts of gaseous hydrogen. In these planets, the H2 molecules are subjected to compressive pressure many hundreds of times higher than that found in the Earth's atmosphere.
"If the two spin isomers were distributed uniformly in gas giants, important conclusions about the magnetic fields of these planets and their stability could be derived. However, in our study we have now succeeded for the first time in demonstrating that ortho- and para-hydrogen are destabilized by extremely high compression pressure. Their respective characteristic properties are lost at around 70 gigapascals. This evidence can significantly expand our understanding of quantum mechanical processes," says first author and physicist Dr. Thomas Meier from the University of Bayreuth.
More information: Thomas Meier et al. Nuclear spin coupling crossover in dense molecular hydrogen, Nature Communications (2020). DOI: 10.1038/s41467-020-19927-y
Journal information: Nature Communications
Provided by University of Bayreuth