Large droplet sorting now possible at high speeds

Bioengineers from UCLA and University of Tokyo have significantly increased the speed at which large liquid droplets, potentially containing individual live cells, can be sorted intact and in bulk.
According to research published in Science Advances, the advance could lead to faster screening —20 times faster than currently available technologies—for products manufactured by cells, such as biofuels or antibodies.
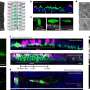
Droplet microfluidic technologies have become powerful tools in medicine and biotechnology. Inside these microfluidic devices are tiny lanes that help route millions of fluid drops, which act as miniature test tubes to grow cells and foster chemical reactions. Drops that contain unique growth or reactions can be automatically sorted to isolate cells of interest from the rest.
Smaller droplets, about a third of the thickness of a human hair in diameter, have been previously used to grow or react cells for a few hours. The smaller mass of these droplets makes them easier to sort at high rates by instruments.
But the droplets are not large enough to allow growth and long-term survival of most cells. Increasing the diameter of a droplet slightly more than two-fold results in 10 times as much volume, enough to completely envelop a cell with an ample cushion. That liquid cushion can keep cells alive much longer and even allow them to grow and divide inside the droplet.
However, these larger droplets break apart during processing in previous systems, mostly due to their higher inertia as they were moved around.
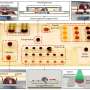
"Up until now, the slower speeds required to move these larger droplets diminishes many of the advantages of using microfluidics, so we set out to change that," said Dino Di Carlo, UCLA's Armond and Elena Hairapetian Professor in Engineering and Medicine and one of the senior authors on the study. "The key in this research is: instead of applying a very strong electric field all at once to move and sort a droplet, which usually rips the droplets apart, we apply a much smaller electric field around each droplet many times in a sequential manner, to slowly divert it from its path. Imagine deflecting a balloon with many small fans all aligned and synchronized to blow only when the balloon passes versus using one large wildly turbulent fan."
he researchers demonstrated the technique using large droplets with a variety of cells, including cancer cells, stem cells, microalgae and yeast. They found the new methodology could keep cells alive, growing and secreting biological products longer in the larger droplets and allow the team to sort the cells at much higher speeds. The advance brings automated sorting and analytical processing speeds of large droplets in line with that of smaller droplets.
"This opens up some new avenues in biologic and cell therapy manufacturing, precision medicine, regenerative medicine and green biotechnology," Di Carlo said. "For example, we can now incubate and grow all kinds of cells and then use microfluidic processing to search for cells with particular important growth or production traits. This could include finding the most promising T cells to fight cancers, or cells that secrete antibodies for infectious diseases such as COVID-19."
Di Carlo said the technique could apply to agriculture-based biotechnologies because the system could sort algae needed for biofuels or vitamin production at faster rates.
More information: A. Isozaki et al. Sequentially addressable dielectrophoretic array for high-throughput sorting of large-volume biological compartments, Science Advances (2020). DOI: 10.1126/sciadv.aba6712
Journal information: Science Advances
Provided by UCLA Samueli School of Engineering