Biocompatible gold nanoparticles will accelerate individual cancer diagnosis and treatment

An international group of researchers from NUST MISIS and Clemson University (Clemson, U.S.) has proposed a new method for producing gold nanoparticles based on synthesis under the influence of ultraviolet radiation. The method excludes the use of aggressive chemical agents and the obtained nanoparticles are safe for the body and can be used for the diagnosis and treatment of cancer. The results are published in the international scientific journal Biomaterials Science.
Cancer remains one of the most common causes of death in the world. Therefore, researchers do not stop looking for ways to diagnose and treat cancer, including the ones connected with the use of more and more popular nowadays nanotechnologies.
Gold nanoparticles are used in the process of catalysis, in electronics, solar cells, but they are of greatest interest for biomedicine. Their important advantage is a combination of properties necessary for the so-called bioimaging, that is, a detailed diagnosis of the tumor and subsequent therapy.
Gold nanoparticles are commonly used as bioimaging agents in computed tomography. Tumor therapy using gold nanoparticles can be carried out by the so-called photothermal therapy, when the particles first accumulate in the tumor and then heat up under the influence of an external field and destroy cancer cells.
Existing methods for producing gold nanoparticles usually require the use of fairly aggressive chemical agents, which complicates their further use in biomedicine or require several stages of synthesis, which makes production more expensive.
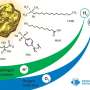
In their work, scientists from NUST MISIS and Clemson University propose a new "environmentally friendly" method for producing gold nanoparticles, in which HAuCl4 gold salt is mixed with a copolymer consisting of polylactic acid-polyethylene glycol in the presence of polyvinyl alcohol and a special Irgacure photoinitiator. New technology eliminates the use of aggressive substances and chemical agents that are toxic to a living organism.
"Despite a long and slightly scary list of components, all of them are highly biocompatible and are actively used in biomedicine," said Roman Akasov, a research associate at the NUST MISIS Biomedical Nanomaterials Laboratory, one of the co-authors of this work. - The resulting mixture is mixed under the action of ultrasound, forming a double emulsion of water-oil-water. Then it can be irradiated with ultraviolet light, as a result of which gold nanoparticles are formed in the solution. In this case, the particles are surrounded by a polymer, which gives them the properties of biocompatibility and stability in aqueous solutions. In this case, the emulsion turns from whitish-transparent to red, which is an indicator of successful photopolymerization. The particle size in our experiments was about 100 nanometers, which is suitable for biomedical applications, and the particles were not toxic to the cells."
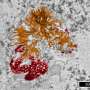
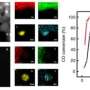
In the work, the authors were also able to show that gold nanoparticles accumulate in the cytoplasm of cells, both tumor glioma and immune cells—macrophages. This opens up the possibility of individual diagnosis and therapy of tumors. In the future, it is planned to modify the surface of nanoparticles with special molecules to find a tumor in the body. However, researchers offer another option for using their method—as a bioconstructor.
The obtained emulsions can be introduced into the cell or even the body even before the photopolymerization stage (the process of polymer synthesis under the influence of light) and synthesized into gold nanoparticles directly in the tissue under study. Moreover, the properties of the obtained nanoparticles will make it possible to analyze the features of the living environment in which they are located, which can be an important tool for studying the biology of the cell and the processes that occur in it.
Currently, the group continues a series of laboratory experiments as part of the preclinical phase of research.
More information: Mohamed F. Attia et al. In situ preparation of gold–polyester nanoparticles for biomedical imaging, Biomaterials Science (2020). DOI: 10.1039/D0BM00175A
Journal information: Biomaterials Science