Scholes finds novel magnetic field effect in diamagnetic molecules
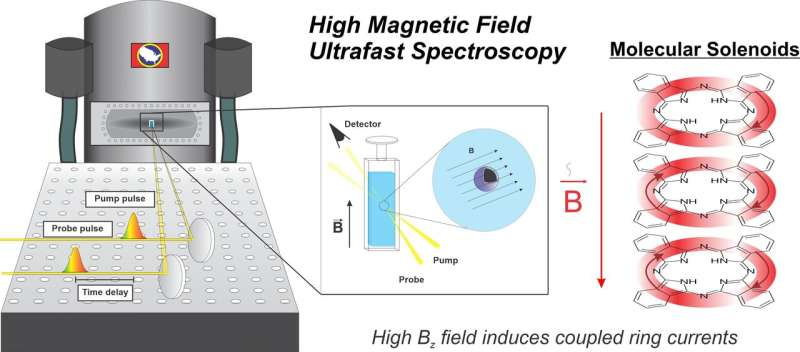
The Princeton University Department of Chemistry publishes research this week proving that an applied magnetic field will interact with the electronic structure of weakly-magnetic, or diamagnetic, molecules to induce a magnetic-field effect that, to their knowledge, has never before been documented.
With the experimental application of magnetic fields up to 25 Tesla, molecules with little intrinsic magnetism exhibit magneto-sensitive optical and photophysical properties, according to the paper, "Ring currents modulate optoelectronic properties of aromatic chromophores at 25 Tesla," published in the Proceedings of the National Academy of Sciences (PNAS).
Gregory Scholes, the William S. Todd Professor of Chemistry, and Bryan Kudisch, a fifth-year graduate student and the paper's lead author, said the discovery could allow scientists to fundamentally change the electronic and photophysical properties of some classes of molecules by using the magnetic field as a "handle."
Experimenting with a magnetic field almost 1M times stronger than that of the Earth, researchers in the Scholes Group were able to modify the optoelectronic properties of model nonmagnetic organic chromophores. The modifications, according to the paper, arise from the induction of ring currents in the aromatic molecules.
"No one would expect an organic molecule with no metal and no intrinsic magnetism to have such an obvious magnetic field effect," said Kudisch. "We're using some of the biggest magnetic fields generated on earth, that's fair. But at the same time, we're seeing something that's never been seen before. And then to come up with a suitable explanation invoking a magnetic field effect commonly seen in nuclear magnetic resonance (NMR), namely aromatic ring currents, is very fulfilling."
Aromatic ring currents can be understood as the proposal that electrons delocalized by aromaticity will move circularly when a magnetic field is applied perpendicular to the aromatic plane, typically nudging chemical shifts of nearby atoms in NMR spectroscopy.
"This research shows that this is a phenomenon with very real chemical implications," Kudisch added. "Here, we've taken something that is commonplace in one type of spectroscopy and have shown how it morphs in a completely unexpected way while using our spectroscopic methods."
For the experiment, researchers chose a model aromatic chromophore called a phthalocyanine, which has a molecular structure similar to chlorophyll—nature's light absorber—but with stronger absorption of visible light and higher stability. The calculations on this model phthalocyanine compound and its aggregates showed clear, magnetic field-dependent changes to phthalocyanine's ability to absorb light. These results mark the first to demonstrate magnetic field dependent changes to the absorption spectrum of diamagnetic molecules. But it wasn't until researchers applied the classical analogue of the solenoid that the experiment sharpened into clarity.
A solenoid is an electromagnetic device that effectively converts electrical and magnetic energy using conductive loops of wire arranged like a spring. With their thinking grounded in the behavior of solenoids, Kudisch said, they were able to rationalize that the increased magnetic field sensitivity they were observing in the phthalocyanine aggregates could depend on the relative arrangement of the phthalocyanine rings in the aggregate.
"Not only did this add extra validation to our computational support, but it also lent credence to this idea of coupled aromatic ring currents—the ring currents of neighboring phthalocyanine chromophores in the aggregate have a geometry dependent on amplification of magnetic field sensitivity," said Kudisch. "Just like the solenoid."
Initiated three years ago, the research project combined experiments using a high magnetic field and ultrafast spectroscopy capabilities. Part of it was performed with the Split-Florida Helix Magnet at the National High Magnetic Field Facility in Tallahassee, Florida, which boasts the world's strongest magnet for NMR spectroscopy. This one-of-a-kind magnet can reach and sustain magnetic field strengths of up to 25 T in a completely resistive fashion—in and of itself likely the most powerful solenoid on the planet. When operational, the magnet uses 2% of the power in the city.
Scholes noted that the PNAS paper marks his group's second publication from work using the Florida split-helix magnet, a collaboration that started over eight years ago when the magnet was being designed. His group's role was to propose and design the ultrafast laser system that connects to the magnet.
"It's relatively easy to get such high magnetic fields on an NMR magnet, but our experiments require you to get light and shine it onto the sample and then to get that light out somehow. And for that, we needed the lab in Tallahassee. It's a bunch of near-impossibilities coming together," said Kudisch.
Kudisch said that obtaining phthalocyanine aggregates in the form of organic nanoparticles for their experiments was "the simplest part," owing to previous collaborations with Princeton's Department of Chemical and Biological Engineering. Other collaborators on the paper include Milan Polytechnic, and the National University of Cordoba.
Overall, he said, the "eclectic" atmosphere of the investigations in the Scholes Lab contributed to the success of the project.
"The context is, this lab is thinking about some of the most pressing problems in physical chemistry that no one's thought about and figuring out if the ideas we come up with are testable," said Kudisch. "When you really dive down into it, what we're interested in is how deep into the rabbit hole of ultrafast spectroscopy we can go, and what it can allow us to learn in a variety of different fields."
More information: Bryan Kudisch el al., "Ring currents modulate optoelectronic properties of aromatic chromophores at 25 T," PNAS (2020). www.pnas.org/cgi/doi/10.1073/pnas.1918148117
Journal information: Proceedings of the National Academy of Sciences
Provided by Princeton University



















