Same genes, same conditions, different transport
The bacterium Lactococcus lactis, which plays an important role in the dairy industry, is unable to produce the amino acid methionine and has to rely on uptake from the environment. To do this, the bacteria have two systems with high and low affinity. University of Groningen microbiologists discovered that cells growing in a clonal population can differ in the uptake system they use. Furthermore, the choice for either system is maintained over many generations. It is the first time that such stable heterogeneity is observed in an amino acid uptake pathway. The results were published in the journal Nature Communications on 5 March.
It has long been known that bacteria grown in a clonal population (where all cells share the same genes) can nevertheless behave differently. Under harsh conditions, some cells may go into survival mode by sporulation while other cells do not. "It is a kind of bet-hedging," explains University of Groningen Professor of Molecular Genetics Oscar Kuipers. "Sporulation is costly for a cell, and conditions may improve. Or they may not. Taking both routes ensures that part of the population will always survive."
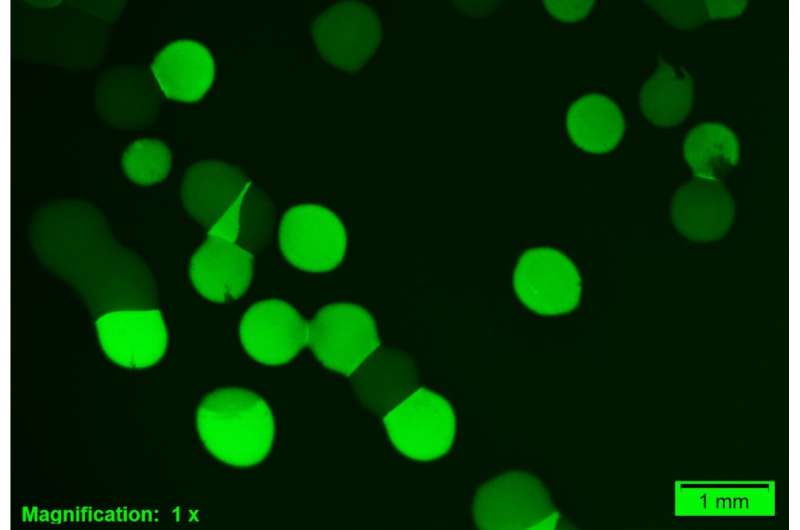
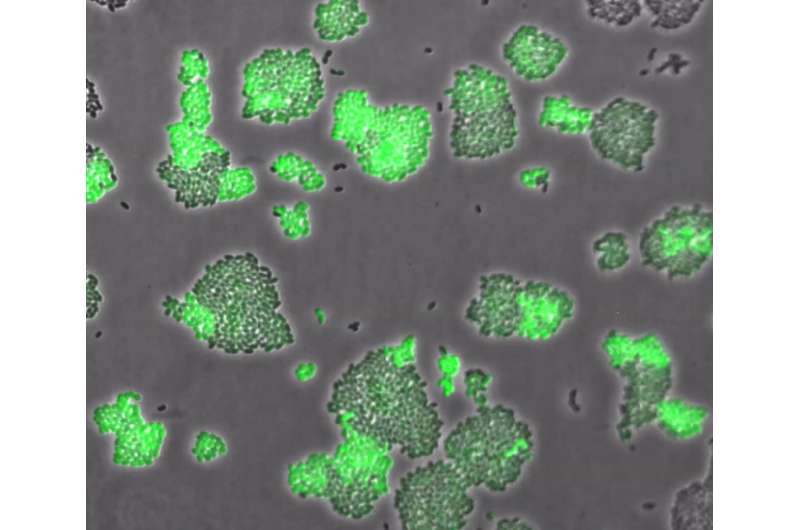
However, this strategy had never before been observed in amino acid transport. Under Kuipers' supervision, Ph.D. student Jhonatan Hernandez-Valdes grew Lactococcus bacteria in medium with different methionine concentrations. He added a reporter gene to the cells that made them light up fluorescent green when the high-affinity methionine transporter was expressed. However, under low methionine conditions, he noticed that some cells did not light up.
At first, Hernandez-Valdes thought his cell culture was contaminated. But after several checks, it turned out that all cells carried the green fluorescence gene—though not all of them expressed the high-affinity methionine uptake system. Kuipers: "We discovered that the switch to the high-affinity system is very slow. You could say that the cells are hesitant to switch."
Bet-hedging
Further exploration revealed that something known as a T-box riboswitch was responsible for heterogeneity in the expression of the high-affinity transporter. Apparently, using low or high-affinity transporters makes no difference in terms of methionine uptake. "It appears to be a matter of chance which route a cell will take. But once the choice is made, it remains fixed for over 10 generations."
Cells with either uptake system grow equally well under low-methionine conditions. So why the heterogeneity? Kuipers and his colleagues suggest two potential explanations. The first is bet-hedging, a well-known phenomenon in both bacteria and higher organisms. "Not investing in the high-affinity transport system may offer some advantage under natural conditions." Some cells within the population gamble on an increase in methionine availability, while others take the safe route and switch on the high-affinity system that provides them with the methionine they need even when there is very little around.
"The other possibility is that there is a division of labor: If the two sub-populations cooperate, for example, cells may start to excrete methionine, or die and fall apart, thus making their amino acids available for other cells," Kuipers explains. But this scenario is very speculative.
"We do know that heterogeneity is a good thing for bacteria," he continues. There may be hundreds of different strains in a gram of soil, all battling for their niche. "By splitting into two populations, you can better anticipate changes in the environment. That is always a smart strategy."
More information: Jhonatan A. Hernandez-Valdes, Jordi van Gestel, Oscar P. Kuipers: A riboswitch gives rise to multi-generational phenotypic heterogeneity in an auxotrophic bacterium. Nature Communications, 5 March 2020. DOI: 10.1038/s41467-020-15017-1
Journal information: Nature Communications
Provided by University of Groningen