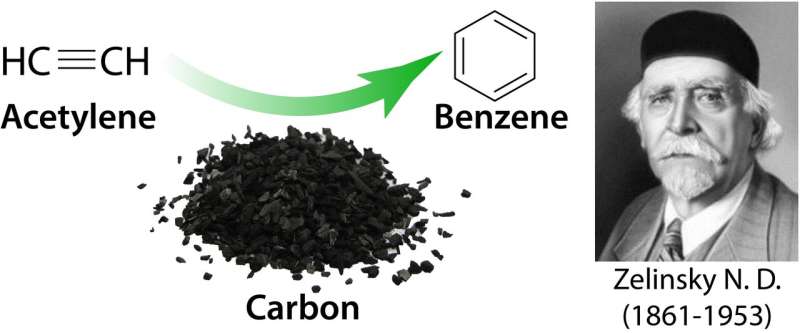
The famous Zelinsky process unveiled: Self-promoted acetylenic cascade produces benzene
Joint computational and experimental study sheds light on a unique property of 2-D materials—dynamic changes of electronic configuration during transformations at the molecular level. Discovery of that mechanistic channel of the Zelinsky reaction allows scientists to design efficient carbocatalytic processes and guides construction of new generation of carbon materials.
In the 1920s, the famous Russian chemist Nickolay Zelinsky was working on development of a convenient route to benzene. Trimerization of acetylene into benzene is usually catalyzed by noble metals and their alloys. However, Zelinsky studied formation of benzene from acetylene in the presence of charcoal and managed to obtain good yields. The process was widely recognized, but its mechanism, very fine and complex, remained a puzzle for decades.
The process investigated by Zelinsky was one of the first examples when traditional metal catalysts were replaced with carbon to catalyze the reaction. Within current state-of-the-art concepts, the core process is today called carbocatalysis, and its advantages include simplicity, sustainability, cost-efficiency and practical use.
In spite of the seeming simplicity of the reaction, the exact catalytic channel to assemble benzene from three acetylene molecules is unknown. As with many carbocatalytic processes, the catalytic channel of the Zelinsky process remained unknown and incomprehensible. Indeed, such chemical transformations are expected to have a paramount level of complexity caused by involvement of fluxional (metastable) active centers on the carbon surface.
A chemical channel of Zelinsky process was unveiled by computational modeling and experimental verification. The key to understanding the mystery of Zelinsky reaction turned out to be carbene active centers localized on carbon atoms at the zigzag edge of graphene-type material.
Quantum chemical calculations of the reaction mechanism provide reconstruction of free energy profiles and visualize spatial distributions of spin density for each stage of the acetylene cyclotrimerization reaction. It was found that stepwise addition of acetylene molecules to the catalytically active center is promoted by continuous spin migration onto β-carbon of the added C2 unit. Furthermore, reversible spin density oscillations facilitate the product formation and regenerate the catalytically active centers. At the same time, the free energy driving force ensures the preference of cyclotrimerization over linear oligomerization after the addition of three acetylene species.
The overall catalytic cycle was governed by reversible spin density oscillations. These oscillations were responsible for the generation of the product and re-activation of the catalytically active centers. The observed phenomenon represents a unique property of the π-electron-conjugated 2-D carbon systems.
Apart from their fundamental importance, carbocatalytic reactions gain a lot of attention for environmental and sustainability reasons. The key issue in this regard is to renounce the use of transition metal catalysts, especially those with noble metals. Their high costs, along with the unavoidable leaching of toxic metal-containing species, represent well-known drawbacks. In contrast to the metal catalysts, their carbon counterparts are cheap and non-toxic.
The article, "Carbocatalytic Acetylene Cyclotrimerization: A Key Role of Unpaired Electron Delocalization," was published in the Journal of the American Chemical Society.
More information: Evgeniy G. Gordeev et al. Carbocatalytic Acetylene Cyclotrimerization: A Key Role of Unpaired Electron Delocalization, Journal of the American Chemical Society (2020). DOI: 10.1021/jacs.9b10887
Journal information: Journal of the American Chemical Society
Provided by Zelinsky Institute of Organic Chemistry