A study of catalysts for synthesizing ultra-high molecular weight polyethylene
A chemist from RUDN University as a part of a team of researchers has synthesized new highly active catalysts for the production of ultra-high molecular weight polyethylene, which is necessary in the production of bulletproof vests, pipes, parachutes, prostheses and much more. The use of the new catalysts will significantly reduce the production cost of this type of polyethylene. The article was published in the journal Inorganica Chimica Acta.
Ultra-high molecular weight polyethylene (UHMWPE) is a polymer of linear structure with a molecular weight of 1 million times more than that of water. Due to the ultra-high mass of molecules, UHMWPE has properties that other types of polyethylene lack: high strength, shock and frost resistance, low coefficient of friction and physiological inertness. The high demand for this raw material prompted the Ministry of Industry and Trade of the Russian Federation to name UHMWPE one of the first products on the import substitution list. But the traditional method of producing this polyethylene by gel spinning is too demanding, as it requires a large number of expensive solvents. The industry needs new methods, and the innovative solid-phase method is considered promising.
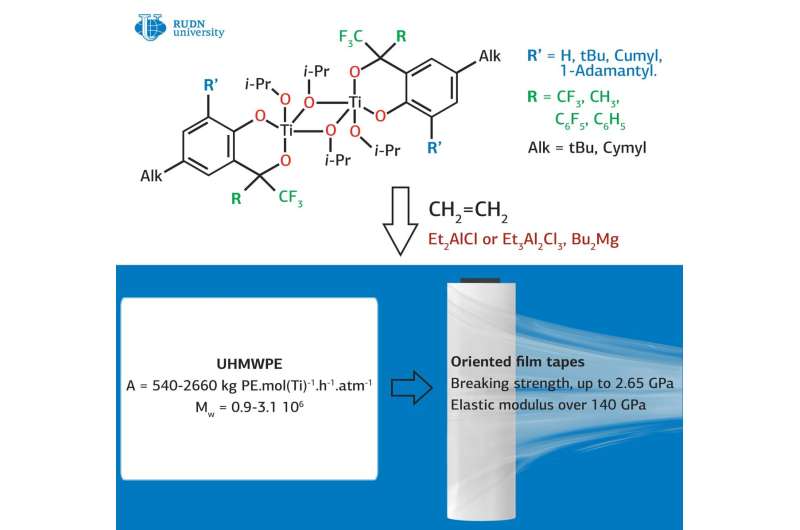
Previously, the researchers suggested using a group of titanium complexes stabilized with 2-hydroxymethylphenol derivatives as catalysts. These substances, according to the researchers, are able to catalyze the synthesis of UHMWPE without solvents. Now Viktor Khrustalev from RUDN University and his colleagues from a number of institutes of the Russian Academy of Sciences and universities of the Russian Federation have developed a new and more efficient version of the catalyst.
The chemists proposed adding ligands (molecular groups) with fluorine compounds rather than hydrogen to the titanium complexes, as it was in the previous version of the catalyst. The replacement of hydrogen with fluorine resulted in an increase in the lipophilicity of ligands and an increase in the acidity of hydroxyl groups.
During the experiment, the researchers found that fluorinated catalysts containing fluorine are superior to non-fluorinated analogues in activity and stereoselectivity during polymerization of polyethylene. The results indicate that the presence of a substituent, regardless of its size, in the ortho position to the hydroxyl group of the phenolic ligand significantly affects the mechanical properties of UHMWPE.
All compounds were isolated with a yield of 62 to 93% in the form of red, air-sensitive powders. Product structures were unambiguously determined by X-ray diffraction study. As a result, synthesis optimization has significantly improved the mechanical strength of UHMWPE, although initially this was not the main goal of the study.
In the future, the chemists plan to further explore the effect of other structural fragments on the activity of catalytic systems and the properties of the obtained polymers.
More information: Vladislav A. Tuskaev et al. Novel alkoxo-titanium(IV) complexes with fluorinated 2-hydroxymethylphenol derivatives as catalysts for the formation of ultra-high molecular weight polyethylene nascent reactor powders, Inorganica Chimica Acta (2019). DOI: 10.1016/j.ica.2019.119159
Journal information: Inorganica Chimica Acta
Provided by RUDN University