Ancient origins of allosteric activation in the oldest kinases
One of the key features in the evolution of more complex organisms is the emergence of allosteric regulation. Allostery is a process by which a protein's activity can be modulated by binding an effector molecule distal to the active site.
Despite the enormous importance of allostery in biology, the question of how such a feature evolved is unexplored territory.
In an article published online on February 22 in Science, professor of biochemistry and Howard Hughes Medical Institute Investigator Dorothee Kern and her lab address what is arguably one of the most fundamental evolutionary drivers for biology—allostery.
By tracing the evolutionary path of modern protein kinases from their ancient common ancestors about 1.5 billion years ago to the present, Kern and her colleagues discovered the ancient origins of allosteric regulation for the first time.
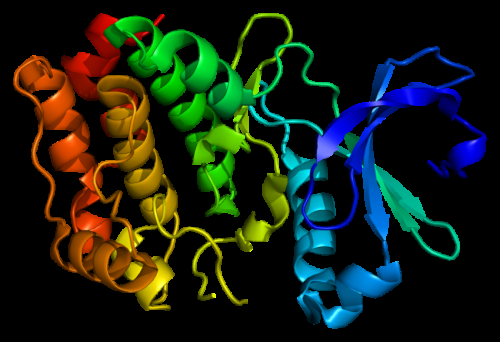
To study such a fundamental question, the researchers chose to resurrect the evolution of Aurora kinase together with its allosteric regulator, TPX2. These proteins control the cell cycle in humans and are therefore hot cancer targets.
In the paper, the scientists first calculated the amino acid sequences of these ancient proteins using the hugest sequence database available to date and bioinformatics. They then made these enzymes in the laboratory and characterized their biochemical properties.
They found that the oldest kinases (about 1.5 billion years old) already use autophosphorylation for their regulation. This makes sense from an evolutionary point of view since the process needs only its own catalytic machinery.
The more sophisticated allosteric regulation, via binding to a second protein, starts about 1 billion years ago with the occurrence of that partner, TPX2.
Strikingly, the scientists found that contrary to the common view, there is no coevolution—reciprocal changes in both partners along the evolutionary trajectory—but that rather the entire interphase of their interaction stays constant for 1 billion years. In other words, they found that co-conservation was an extremely strong evolutionary constraint.
But what happened to allosteric activation? This advanced regulation is gradually evolving over 1 billion years leading to the strongest allosteric activation in our human kinase. The researchers discovered that its mechanism is the evolution of a sophisticated allosteric network that spans the entire kinase from the site of the TPX2 binding to the other side of the protein.
Kern's findings have far-reaching implications for understanding the evolution of complexity from extremely primitive creatures to the human species, and for novel approaches to cancer therapy taking advantage of the newly discovered allosteric networks in our modern proteins.
More information: Adelajda Hadzipasic et al. Ancient origins of allosteric activation in a Ser-Thr kinase, Science (2020). DOI: 10.1126/science.aay9959
Journal information: Science
Provided by Brandeis University