Microswimmers swim faster over slippery surfaces
Tiny self-propelling spheres, measuring only micrometers, move faster over a hydrophobic silicone surface than they do over hydrophilic glass. "Almost nobody had realized that the substrate matters," says Stefania Ketzetzi, the researcher who discovered the effect, researched it and explained it. She publishes about it in the journal Physical Review Letters.
"This was an accidental discovery," says Ketzetzi, a scientist in the Daniela Kraft group, who researches microswimmers. These are tiny colloidal spheres measuring only micrometers. One half of the sphere is covered in a thin layer of platinum.
Tiny rockets
When these particles are suspended in a hydrogen peroxide solution, the platinum acts as a catalyst. It promotes the chemical reaction of hydrogen peroxide into water and oxygen. The formation of reaction products on one side of the particle creates a fluid flow that propels the particle like a tiny rocket. Because of their self-propulsion, they are called 'active' particles. One day, they may be applied as motor parts for micro-robots.
"I stumbled upon the substrate effect while working on a project for which I needed to use polymer-coated glasses as substrates," says Ketzetzi about the publication in Physical Review Letters. "Those substrates were highly hydrophilic. I noticed that the same swimmers were moving much slower. Why?" Together with group leader Kraft, she decided to investigate.
Silicones
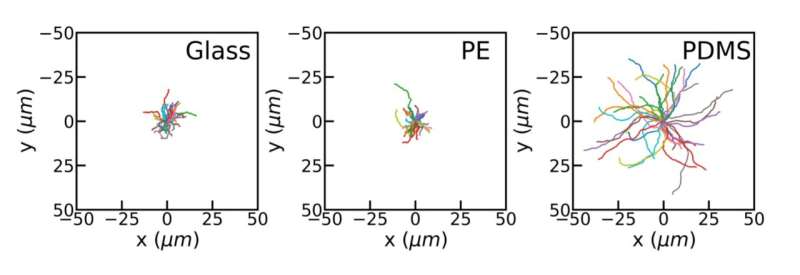
The researchers checked systematically whether the electrical charges of the substrates mattered for the speed, but this did not yield a clear result. The roughness of the substrates also did not yield a correlation. Then, they decided to test substrates with varying hydrophobicity: the hydrophobic polydimethylsiloxane ('silicones') versus glass and hydrophilic polyethylene. It turns out that the slipperiness of the substrate makes a difference for the propulsion speeds: the tiny rockets reached a speed of 2.8 micrometers per second on silicones, versus 1 micrometer per second on the other substrates.
A measure of hydrophobicity is the contact angle, the angle that the surface of a water drop makes with a solid surface. On hydrophilic substrates, the drops spread widely and the contact angle is small. On hydrophobic substrates, the contact angle is larger, typically more than 90 degrees. The researchers found that for similar contact angles, the speeds were also similar, while a higher contact angle also means higher speed.
An explanation was found with help from theoretical physicist Joost de Graaf of Utrecht University. The contact angle of the solution on the substrate relates to the slipperiness of the substrate. The larger the contact angle, the more slippery the substrate, and so the easier for the fluid to flow along it.
Slipperiness
"It turns out that the slipperiness of the substrate makes a difference for the propulsion speeds," says Ketzetzi. The propulsion comes from flows of fluid around the particle. When a particle moves over a substrate, part of these flows will flow between the particle and the surface. This will be easier on slippery, hydrophobic surfaces.
On the other hand, Ketzetzi says, flowing over hydrophilic surfaces, water feels more resistance, so the propulsion flows have a harder time moving between the particle and the surface. This will hamper the propulsion of the colloid, which results in slower swimmers over hydrophilic surfaces.
The next step, Ketzetzi says, is to investigate how to use this. One could think of applications like lab-on-a-chip, a miniature chemical lab, or of drug delivery, where drugs are targeted and delivered to particular locations in the body. Ketzetzi: "In applications swimmers will have to self-propel in complex environments with confining walls and obstacles. It is important to understand the effect that these have on the swimmer. Using this new knowledge, we can understand and possibly control the behavior of the microswimmers."
More information: Stefania Ketzetzi et al. Slip Length Dependent Propulsion Speed of Catalytic Colloidal Swimmers near Walls, Physical Review Letters (2020). DOI: 10.1103/PhysRevLett.124.048002
Journal information: Physical Review Letters
Provided by Leiden University