Gut feeling: A network approach towards understanding IBD
Diseases of the gut are often notoriously difficult to diagnose, nevermind understand, which is where a network-based approach comes in. Rather than trying to understand individual symptoms, or the role of individual genes, we can use computational methods to get that deeper understanding—looking at how genes work together in complex systems, and how these fail in a disease.
The Korcsmaros Group at EI and QIB have therefore pioneered a combined experimental and computational pipeline that tells us what genes are the important 'master regulators' in the gut, and which of these genes overlap with those associated with IBD, suggesting new angles to research in the future.
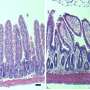
Much of this requires us to look at the cells in the gut. Paneth cells release antimicrobial peptides to protect the gut from pathogens and goblet cells release mucus to create a protective barrier. Dysregulation of their function is implicated in diseases such as IBD.
A promising method of understanding the role of these cells has recently been introduced. Known as organoids, stem cells can be cultured from mice models or from patients to make a small model of a human gut, and the effects of different treatments can then be explored, opening avenues for precision medicine, among others.
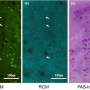
These organoids can also be modified to study the relative importance of different cell types within the system, and to explore interactions between different genes in healthy or diseased cells. One question that scientists have had is whether this 'enrichment' of organoids, to give us more Paneth cells or goblet cells for example, gives us a realistic model.
The good news, according to Ph.D. student Agatha Treveil of EI, who first authored the paper, is that: "Based on our transcriptomics analysis, we see clear increases in targeted cell types as we expected from using the organoid enrichment methods."
The pipeline, published in Molecular Omics, allows us to compare gene expression in organoids that are enriched for Paneth cells or goblet cells with those that are not. These can then be compared with a growing database of known gene interactions.
Essentially, we can tag the genes which regulate others—highlighting in particular those that regulate lots of processes and are important for goblet and Paneth cell function. The finding that many of these genes overlap with inflammatory processes and IBD both confirms the important role of Paneth cells in IBD, and opens another route to understanding this range of costly illnesses.
Agatha Treveil continues, "this is a pioneering step forward in establishing a pipeline to better understand the gene networks at play in the gut. It enables us to design new experiments to explore IBD-related processes further."
Dr. Tamas Korcsmaros, the leader of the project, said, "This project nicely represents the collaborative environment we have at the Norwich Research Park, with UEA, EI and QIB focusing on new experimental models, transcriptomics and computational approaches, and then applying them in translational projects with clinical researchers of the Norfolk and Norwich University Hospital. Now, we have a pipeline that we can use to investigate organoids generated from IBD patients, and analyse drug compound or probiotic effects on these key cell types."
More information: A. Treveil et al, Regulatory network analysis of Paneth cell and goblet cell enriched gut organoids using transcriptomics approaches, Molecular Omics (2019). DOI: 10.1039/c9mo00130a
Provided by Earlham Institute