Fishing for novel cellulose degraders
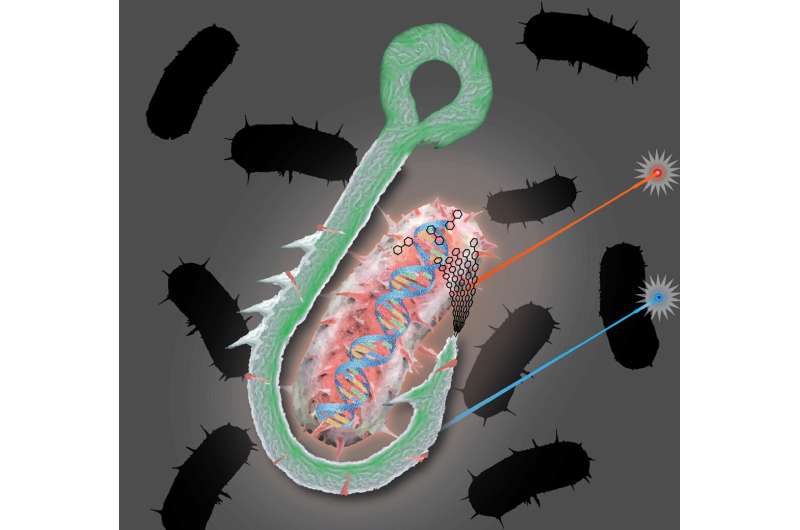
One of the most vital pieces of equipment for fly fishing is a boxful of lures. Designed with feathers or wires to mimic an insect or a particular movement, each of these lures are the bait designed to attract specific catches. A similar technique has been developed by researchers led by Tanja Woyke at the U.S. Department of Energy (DOE) Joint Genome Institute (JGI), a DOE Office of Science User Facility located at Lawrence Berkeley National Laboratory (Berkeley Lab). Using a single-cell screen, they can now identify microbes with specific functional characteristics. When they tested the screen method on a microbial community from geothermal hot springs, they uncovered a novel cellulose-degrading bacterium typically found in low abundances.
Microbes are known to be key contributors to the planet's biogeochemical cycles, but the vast majority remains uncultivated and unknown. The challenge remains to learn more about what microbes are present in any given ecosystem—particularly those that are in low abundances—and their functional roles in their community. This study shows how the function-driven, single-cell screen can uncover more about cellulose-degrading microbes. The new technique is another tool for microbial ecologists to learn more about the diversity of microbial enzymes, which could have a plethora of potential applications to industrial processes.
JGI researchers developed a function-driven, single-cell screen that they described as a "hook and bait" approach to isolate cellulose-degrading microbes. They first bound single microbes to fluorescently labeled cellulose particles that served as the "bait," and then they isolated the particles through the "hook" of fluorescence-activated cell sorting to sort cells into several small pools, followed by whole genome amplification and shotgun sequencing. The team first tested the approach on bacterial laboratory models to set a reference for its performance, and then applied it to environmental samples from the Great Boiling Spring Geothermal Field in Nevada. To validate the predicted cellulases from their captured genomes, the team then used DNA synthesis and cloning, followed by protein in vitro expression and mass spectrometry-based enzyme activity assay.
While the screen caught several microbes from known cellulose-degrading groups, the researchers also hooked and were able to reconstruct a high-quality draft co-assembled genome for a new genus level member of the novel candidate phylum Goldbacteria. They've proposed the name Candidatus Cellulosimonas argentiregionis for this microbe.
The work is an example of the application of function-driven genomics, which Woyke and her colleagues define as "any form of functional enrichment leading to the sequencing of genomes from either individual cells or consortia of cells," The approach can be further developed to bait for specific functions other than cellulose-degradation to tease out uncultivated microorganisms of interest from complex environmental samples.
More information: Devin F. R. Doud et al. Function-driven single-cell genomics uncovers cellulose-degrading bacteria from the rare biosphere, The ISME Journal (2019). DOI: 10.1038/s41396-019-0557-y
Journal information: ISME Journal
Provided by DOE/Joint Genome Institute